A new RCT of curosurf given by nebulisation was published shortly after a systematic review of the previous data. The RCT showed no real benefit, whereas the SR suggested efficacy.
The RCT first, this international trial (Dani C, et al. A Randomized, Controlled Trial to Investigate the Efficacy of Nebulized Poractant Alfa in Premature Babies with Respiratory Distress Syndrome. J Pediatr. 2022) randomized 129 babies (28 to <33 weeks GA) into 3 groups, 2 received different doses of Curosurf nebulised by a special device that could be attached to the nasal prongs (either 200 or 400 mg/kg). Eligible babies were those with HMD who needed between 25 and 40% oxygen on CPAP 5 to 8 cmH2O.
The babies in the 3rd group stayed on regular CPAP. The primary outcome variable was needing intubation; for surfactant therapy (over 40% oxygen need) or becoming hypercapnic (pCO2>60 with pH<7.2) or having lots of apneas.
The study was stopped for futility after just over 40 babies per group, which, as always, is disappointing. The relative numbers of babies who had primary outcome criteria in the higher dose surfactant group (49%) was less than the controls (58%). The 95% confidence intervals for the relative risk of “respiratory failure” (the primary outcome criteria) was 0.56-1.26 for the 400 mg/kg dose and 0.68-1.42 for the 200 mg/kg dose.
It took about 30 minutes to nebulise the higher dose (and half that for the lower dose) and the babies tolerated it well. Post-hoc subgroup analyses suggest that the nebulised surfactant was more effective in the more mature babies.
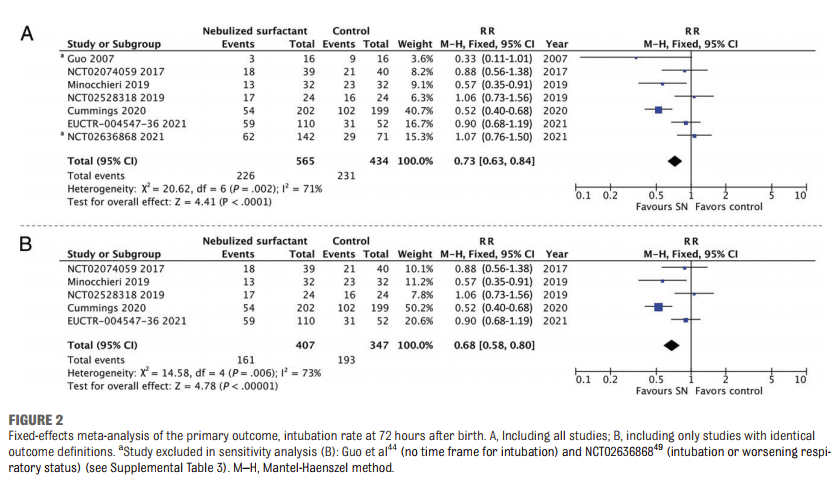
The systematic review (Gaertner VD, et al. Surfactant Nebulization to Prevent Intubation in Preterm Infants: A Systematic Review and Meta-analysis. Pediatrics. 2021;148) included the results of that trial (called Curoneb), the numbers are a little different, which seems to be because the SR mixed the results of the pilot and the full trial, so instead of the 129 babies in Curoneb, they report the 152 babies in the combined trials. As you can see from the following Forest plot, the SR overall suggests that nebulised surfactant decreases intubation within the 1st 72 hours of life, but the results are really completely dependent on the Cummings trial, which showed a major decrease by 48% in need for intubation, without Cummings there seems very little there. The I2 shows lot of heterogeneity, but nevertheless, the 95% compatibility limits of the RR from the results of Curoneb overlap with the summary Relative risk, and there is some overlap with Cummings trial.
If we examine the Cummings trial in detail, (Cummings JJ, et al. Aerosolized Calfactant for Newborns With Respiratory Distress: A Randomized Trial. Pediatrics. 2020) it was a strange study, which included babies from 23 to 41 weeks gestation, who were on any mode of non-invasive support between 1 and 12 hours of age, needing, at first, 25 to 40% oxygen, but then later on in the trial that was changed and babies in 21% oxygen were enrolled. There were no objective criteria in the trial design for intubation, or for failure of treatment, even though intubation for surfactant administration within the first 4 days was the primary outcome and the trial was unblinded.
There were actually more late term/posterm babies in the trial than babies under 27 weeks, and there were as many early term babies as those born at 27 or 28 weeks. The following figure, which shows that information, includes 20 babies (which is what the “combined cohorts” means) who were randomized after extubation having already received surfactant while intubated. As you can see most of the babies were more mature infants, over 30 weeks gestation. The babies received 210 mg/kg of Infasurf, via a modified nebuliser via an oral airway.
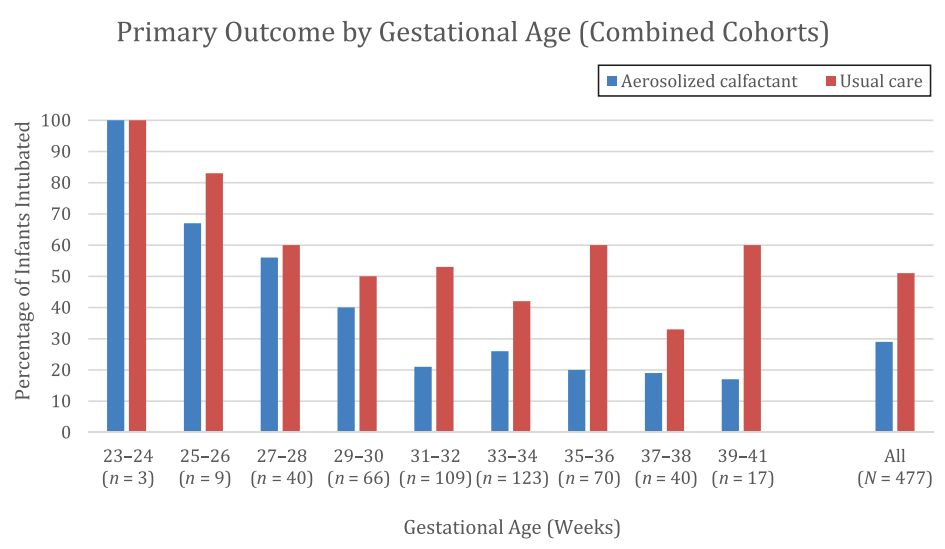
The major problem with this trial, of course, is the use of a subjective outcome criterion in an unblinded study. In addition, the inclusion of full term babies, makes it difficult to generalize the results.
Overall then, it seems that there might be clinical effects of nebulised surfactant, a proportion of the administered dose is deposited in the lungs, and gas exchange tends to improve. In larger preterm babies, this probably somewhat reduces the need for more invasive surfactant administration, but any other benefits (reduced chronic lung disease, improved pulmonary health in infancy, less nosocomial pneumonia or sepsis, improved family interactions or satisfaction) have not been shown.
The implications of all this are that nebulised surfactant has an efficacy which seems limited, and many babies will still need either MIST/LISA or intubation for surfactant therapy. If you take the group that seems to have the best response, between 30 to 34 weeks, a baby that has HMD and is on CPAP with 25 to 40% oxygen in the first few hours, will progress around about half of the time and need surfactant by direct intra-tracheal administration, but if given nebulised surfactant that decreases to between a quarter and a third.
To really prove that you can avoid intra-tracheal surfactant administration in that proportion of such babies really needs another trial, probably with a non-inferiority design. It would be expensive to do this as a masked trial, with a separate team to care for the baby during nebulisation, so I would think an unmasked trial is more likely to be possible, failure criteria should be objective, and it should really be limited to preterm infants.
Is it actually worth doing such a trial? I’m not sure that sometimes avoiding intra-tracheal surfactant administration by nebulising it instead is an outcome that matters enough in larger preterm babies. Laryngoscopy and endotracheal intubation, or passing a catheter through the cords, isn’t much fun for the baby, but the GA group of babies where there seems to be most efficacy have little in the way of serious complications. I think if you could reduce intubations in babies under 28 weeks, then that is a whole different issue, but there doesn’t seem much preliminary evidence that that would work.
Looking at the minor difference in outcomes in the group of 27-28 week babies in the above graph, and that is despite the potential biases in study design, you would need a huge trial to confirm a similar reduction in intra-tracheal surfactant need in the babies for whom it might make a clinically important difference. Unless there are better ways of ensuring that an effective surfactant can be quickly deposited in sufficient quantities in the terminal air sacs, I think we might have to say goodbye to nebulised surfactant as a great idea that didn’t live up to its promise.