I don’t know if there is an “official” definition of prebiotics, but I think of them as molecules present in the diet that promote the growth of probiotic organisms. I believe that originally the term was applied only to molecules that are not digested by humans (or I guess another animal being studied) and are only digested by commensal organisms, including those that have a positive health benefit. I have seen the term used more widely however to include such molecules as lactoferrin which is partially degraded by humans to produce lactoferricin, and which can be absorbed and have direct impacts on health. It also is not, I believe digested by any probiotic organisms.
Although lactoferrin is a very interesting molecule, it probably shouldn’t be considered a prebiotic, the most interesting prebiotic molecules are in fact carbohydrates, including a number of oligosaccharides.
Even though the Human Milk Oligosaccharides (HMOs) are not digested and have no direct nutritional benefit for human babies, they together form the 3rd most abundant component of breast milk, after lactose and lipids. There are very many of them which appear to have importance for the growth of probiotic bacteria.
To get much further into biochemistry than I am really qualified for, the HMOs are a group of compounds which consist of various combinations of glucose, galactose, fucose, N-acetylglucosamine and N-acetyl neuraminic acid. The last mouthful in that sentence is one of a group of molecules, sugars with 9 carbon atoms, which are collectively known as sialic acid; N-acetyl neuraminic acid is also sometimes itself referred to as sialic acid, just to make it a bit more confusing.
This is a wordy preamble to introduce the fact that there is one of the HMOs which looks like it might be extremely important in the pathophysiology of NEC, or rather in the prevention of NEC and that is a molecule known as…. take a deep breath…. Disialyllacto-N-tetraose
Here is schematic of what it looks like

The purple diamonds are the N-acetyl neuraminic acid residues, the blue circle is glucose, the yellow circles are galactose, and the blue square is N-acetylglucosamine. A few years ago now the idea that this particular HMO might be very important arose from a number of studies including an animal model of NEC. (Autran CA, et al. Sialylated galacto-oligosaccharides and 2′-fucosyllactose reduce necrotising enterocolitis in neonatal rats. Br J Nutr. 2016;116(2):294-9), which was followed by a multicentre cohort study (Autran CA, et al. Human milk oligosaccharide composition predicts risk of necrotising enterocolitis in preterm infants. Gut. 2018;67(6):1064-70) showing that among mothers who were provding breast milk to their babies, the infants who nevertheless developed NEC had much lower concentrations of that particular HMO in the breast milk they were receiving.
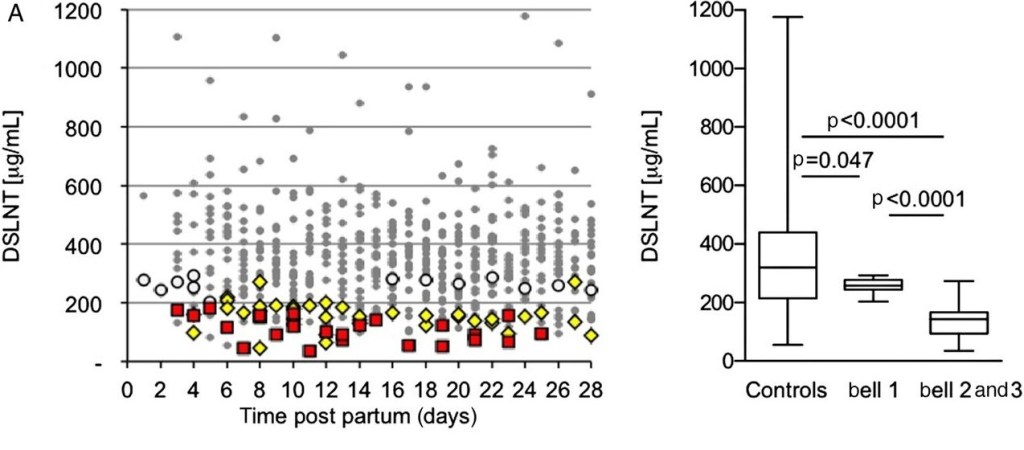
The figure on the left shows cases with Bell stage 3 NEC in red squares, Bell stage 2 as yellow circles and the other grey dots are the matched controls.
This has just been confirmed in an independent cohort, (Masi AC, et al. Human milk oligosaccharide DSLNT and gut microbiome in preterm infants predicts necrotising enterocolitis. Gut. 2020) which again showed much lower DSLNT concentrations in the breast milk of babies who went on to develop NEC. In this study they also analyzed the intestinal microbiome and showed that babies who developed NEC had lower Bifidobacterium longum concentrations.
This work has a number of implications, for one, I wonder whether screening donor mother’s milk for the concentration of DSLNT would be feasible, and whether selecting milk with higher concentrations might enhance the protection that donor milk provides to babies whose mothers cannot produce all the milk they need.
Of course, the question of whether supplementation of the infants’ diet with DSLNT might prevent NEC is going to be the next issue. It appears that it can be synthesised, but I have no idea about the potential cost of synthetic DSLNT or whether it could be extracted from human breast milk.
Prebiotics have actually been tested in clinical trials for NEC prevention, but here you have to be very careful, and realize that not everything that has been tested are actually prebiotics according to the definitions above, and none of the studies have tested any of the most likely effective HMOs, such as DSLNT.
A new network meta-analysis, for example, (Chi C, et al. Effects of Probiotics in Preterm Infants: A Network Meta-analysis. Pediatrics. 2021;147(1)) includes 5 articles that they state studied a prebiotic. One of the studies did not actually include a prebiotic, one of the studies included a group receiving lactoferrin, and the 2 others that are easily available studied inulin. The 5th studied a “fructo-oligosaccharide” which I think is also inulin. None of these molecules are the prebiotics that we need to be studying. This network meta-analysis did, however, report, based on 45 publications including over 12,000 participants, findings that confirm those of other reviews that NEC is reduced by probiotic supplementation and that combination preparations appear to be more effective.
I think the next stage ought to be a trial of babies receiving breast milk (either maternal or donor) and a multi-strain probiotic mixture including B. longum subsp infantis which randomizes the infants to prebiotic or placebo. The prebiotic could either be a mixture of HMOs, or DSLNT, how we can obtain these I am quite unsure.









Pingback: More musings on the microbiome | Neonatal Research
Pingback: AAP guidance on probiotics. As wrong-headed as a head can be wrong. | Neonatal Research
Pingback: Does preterm formula cause NEC? | Neonatal Research
Pingback: NEC awareness day, 17 May 2025. What is new in NEC prevention? | Neonatal Research