We are fortunate in neonatology to not have to worry about C difficile, partly because the name has been changed for this germ as well (now Clostridioides, rather than Clostridium), and it is one less name change to keep up with, but more seriously because it can be devastating, difficult to eliminate and frequently recurring. There are some parallels with NEC, however, including an association with prior antibiotic use, an association with gastric acid production inhibitors, a deranged intestinal microbiome preceding the condition, and a predilection for the most fragile patients. Probiotic prophylactic studies have generally shown efficacy, but one very large trial found no effect (I think because the control group rate was so low), and probiotic prophylaxis is not universal despite a favourable risk-benefit and cost-benefit profile.
There are also differences, including the clear efficacy of an intervention that has not been tried in preterm neonates (to my knowledge): fecal transplantation. And now a trial, just published in the NEJM (Feuerstadt P, et al. SER-109, an Oral Microbiome Therapy for Recurrent Clostridioides difficile Infection. N Engl J Med. 2022;386(3):220-9), of a probiotic preparation specifically designed to prevent recurrence using a mixture of spore bearing Firmicutes, was dramatically successful in reducing recurrence, from 40 to 12%, relative risk 0.32, compatibility intervals 0.18, 0.58. There were zero adverse effects. The role of bile acid metabolism was something I did not know about, but the Firmicutes metabolize primary to secondary bile acids, preventing the C difficile from having access to primary acids, a further analogy with the role of Bifidobacteria in metabolizing HMOs.
With the risk of giving too much press to one specific commercial strain of probiotic for the newborn, another recent publication regarding B longum susbsp infantis (Bajorek S, et al. B. infantis EVC001 Is Well-Tolerated and Improves Human Milk Oligosaccharide Utilization in Preterm Infants in the Neonatal Intensive Care Unit. Frontiers in Pediatrics. 2022;9) looked at stools of 15 VLBW infants before and 15 infants after introduction of probiotic administration in their NICU. None of the control group became colonized with the organism, compared to all of the intervention group, the more heavily colonized the baby was, the less Enterobacteria were present in their stools. The B infantis was very efficient at metabolizing oligosaccharides, leaving practically zero of the measured HMOs in the babies’ stools.
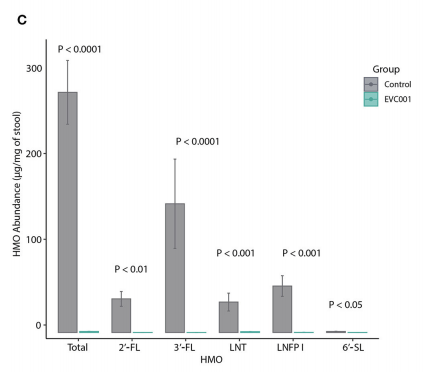
The abbreviations refer to commonly studied HMOs, 2′-fucosyllactose (2′-FL), 3-fucosyllactose (3′-FL), lacto-N-tetraose (LNT), Lacto-N-fucopentaose I (LNFP I) and 6′-Sialyllactose (6′-SL). I don’t know if the LNT is the same thing as the DSLNT (disialyllacto-N-tetraose) that I have previously discussed, but if so it is an extremely important HMO, and the ability to metabolize it may be important for probiotic efficacy.
Antibiotics are well known to effectively kill the probiotic organisms as well as the pathogens against which they are directed. A broad range of molecules (penicillins, macrolides, clindamycin) seems to destroy Bifidobacteria and Lactobacilli, they do this when administered antenatally, with prolonged impacts on the intestinal microbiome (Diamond L, et al. Impact of intrapartum antibiotics on the infant gastrointestinal microbiome: a narrative review. Arch Dis Child. 2021), and of course when administered directly to the infant (Tapiainen T, et al. Impact of intrapartum and postnatal antibiotics on the gut microbiome and emergence of antimicrobial resistance in infants. Sci Rep. 2019;9(1):10635). A recent study from Shanghai randomized 55 full term infants with suspected early onset sepsis receiving 7 days of piperacillin-tazobactam, to either control, 1 week of concurrent probiotics, or 1 week of probiotics after the antibiotics finished. (Zhong H, et al. Impact of probiotics supplement on the gut microbiota in neonates with antibiotic exposure: an open-label single-center randomized parallel controlled study. World J Pediatr. 2021;17(4):385-93). They showed a simplification of the microbiome and near elimination of Bifidobacteria with pipi-tazo when stools were sampled 1, 2, and 6 weeks later. When a probiotic mixture of Bifidobacterium longum, Lactobacillus acidophilus, and Enterococcus faecalis was given after the antibiotics it wasn’t very effective at restoring the microbiome. When probiotics were given concurrently they modified the “devastation” of the microbiome by the antibiotics, allowing a faster recovery, particularly of the Bifidobacteria.
We start probiotics along with the feeds (maternal or donor breast milk) in our extremely and very preterm babies, and I have often wondered whether it was effective to give probiotics with the feeds to those babies who were receiving antibiotics, or whether we should wait until the antibiotics have stopped. Although obviously not directly relevant to our preterm babies with our probiotics and our usual ampicillin/gentamicin treatment, this study suggests that maybe it is worth doing after all.








