It can sometimes take years for major advances in medicine, based on clear evidence of improved outcomes, to lead to shifts in practice. One example is the initial demonstration by Liggins and Howie that antenatal steroids (ANS) improved survival of preterm infants, it took 23 years before the American College of OB/GYN supported their use, and low rates of ANS treatment are evident in the data from trials (that is, even from active academic centres) well into the 1990’s. Sometimes the opposite happens, there is a huge shift in practice, even affecting patients for whom there is no good evidence, when the advantages are questionable, and adverse effects have not yet been ruled out.
Perhaps because of their tardiness in accepting ANS prior to very preterm delivery, our US Obstetric colleagues seem extremely keen to use ANS prior to late term delivery. Remember that, in the pivotal ALPS trial (Antenatal Late Preterm Steroids) although “statistically significant”, there was a relatively modest impact on the primary adverse outcome (death or respiratory disease needing over 30% oxygen or positive pressure), an absolute 3% reduction, which was mostly due to an absolute 3% reduction in need for CPAP/Highflow cannula; there were no deaths in either group. That trial also showed an absolute 3% decrease in NICU admission, and an absolute 9% increase in hypoglycaemia, with an NNT of 33 to prevent an additional baby needing CPAP, and an NNH of 11 to have one more baby developing hypoglycaemia.
This new publication demonstrates how much ANS prior to late preterm delivery has changed since then, and the “spill-over” into use for deliveries at term.

These data from a birth certificate database show an immediate dramatic increase in ANS usage, and ongoing increases in use since then, with the shaded part of the graphs starting in Feb 2016 when the trial was published in the NEJM.
Women with diabetes diagnosed prior to pregnancy were not eligible for the ALPS trial, but that hasn’t prevented a major increase in use of ANS for those women, prior to late preterm delivery. The trial also excluded women presenting at full term.
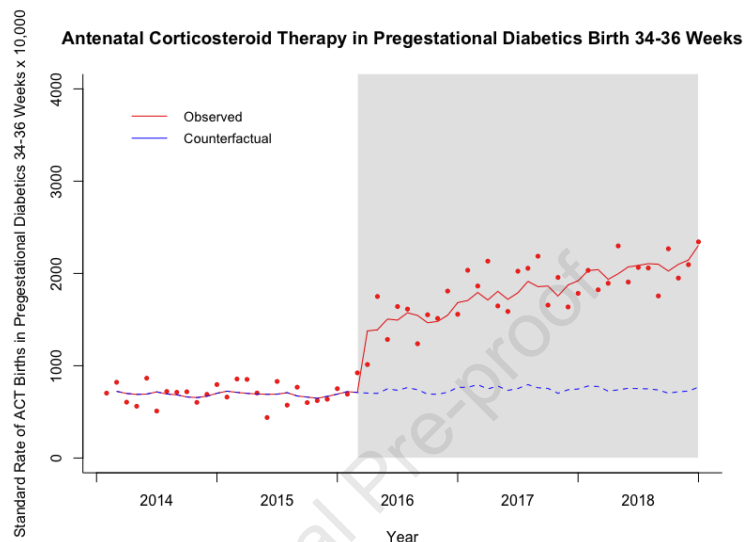
It is really unclear whether ANS are safe for this group of mothers and babies, despite what I said recently about hypoglycaemia, I don’t think we should be trying to create more of it! Particularly among mothers who would not have been eligible for ALPS (term or diabetic) the risk/benefit ratio is completely unknown.
Why ANS might increase hypoglycaemia is studied in another on-line first article in the same journal (Battarbee AN, et al. Mechanism of neonatal hypoglycemia after late preterm steroids: are fetal metabolic effects responsible? Am J Obstet Gynecol. 2022). One would guess that it might be due to endocrine effects, and specifically that betamethasone, which obviously crosses the placenta and has potent glucocorticoid effects in the foetus (that’s why it is being given), would lead to foetal hyperglycaemia and to downstream secondary hyperinsulinaemia. Which is what this study was examining. It was an analysis of data from a subset of babies in the ALPS trial who had umbilical cord blood stored; C-peptide, insulin, leptin, and insulin-like growth factor binding protein 1 (IGFBP-1) were measured in just over 200 babies, about 30% of whom developed hypoglycaemia, 33% in the ANS group and 27% of the controls. The C-peptide, insulin and leptin levels were higher in the ANS babies, and were strongly associated with hypoglycaemia, especially high insulin concentrations above the 90th percentile which had an Odds Ratio for hypoglycaemia of over 6.0.
Should we be giving ANS prior to expected or planned late preterm deliveries? There have been a number of concerns cited and, despite my snide comments about the ACOG earlier on, the current advice regarding late preterm ANS is well written and discusses the potential risks in detail, with a helpful “summary of Evidence” table in an appendix. I must say I disagree with their final conclusion, which is “We recommend offering a single course of antenatal corticosteroids (2 doses of 12 mg of intramuscular betamethasone 24 h apart) to patients who meet the inclusion criteria of the ALPS trial, ie, those with a singleton pregnancy between 34 0/7 and 36 6/7 weeks of gestation who are at high risk of preterm birth within the next 7 d and before 37 weeks of gestation”. They do also recommend, within the document, “against the use of late preterm corticosteroids in pregnant patients with pregestational diabetes mellitus, given the risk of worsening neonatal hypoglycemia”, although the increase in neonatal hypoglycaemia was demonstrated in the ALPS trial which excluded mothers with pregestational diabetes, and therefore applies to all the newborns who are exposed. They have a reasonable discussion of the potential adverse impacts on brain development and they “recommend that patients at risk of late preterm delivery be thoroughly counseled regarding the potential risks and benefits of antenatal corticosteroid administration and be advised that the long-term risks remain uncertain”.
How many obstetricians are really equipped to thoroughly counsel expectant mothers about the possible adverse impact of ANS prior to late preterm birth and the uncertainty of the scientific evidence? How many mothers are equipped to understand and make an informed choice? If I were in that situation (easy to say for someone who could never have been) I would just about be able to comprehend the data and the implications of the data, and that is with a subspecialty in neonatology and years perusing the literature! Perhaps ACOG, and their society for maternal fetal medicine who authored the guidelines, could produce a YouTube video for parents that summarizes the information in a way that most could understand.
If you examine the data from ALPS in detail (the online supplementary appendix) you find that there was no advantage to ANS if the woman was randomized at or after 36 weeks, there was exactly the same incidence of the primary outcome, 7.1%.
The benefit of the steroids was much greater with planned cesarean; 26% primary outcome in controls reduced to 16% with steroids, compared to planned vaginal delivery where the difference was 12.4% to 10.7%. The benefits were also much greater in the group with indicated preterm delivery (induction or cesarean), compared to preterm labour or ruptured membranes, below are the numbers and percentage of the adverse primary outcome according to indication, from the supplementary appendix, the first column of results being the betamethasone group, the second being control, the 3rd is the relative risk and compatibility intervals, and the final figure being the interaction p-value.
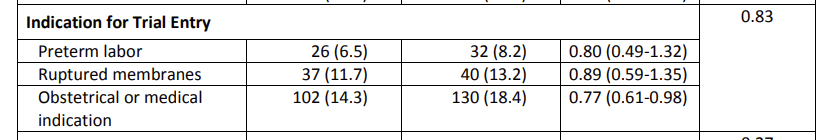
You can see that the absolute reduction in the primary outcome is about 1.5% for the first 2 indications, and about 4% for those with induced labour or planned cesarean deliveries.
The ACOG statement reviews some of the evidence suggesting a possibility of long term impacts on brain development, and comes to no clear conclusion as to whether this intervention is safe or not, which I think is the correct interpretation, there is no clear conclusion.
As I said above, I disagree with their final conclusion that ANS should be offered to all women with increased risk of preterm delivery at 34 to 36 6/7 weeks. The evidence of benefit at 36 weeks is lacking, and those babies are just being exposed to an increase in hypoglycaemia and the unknown long-term effects without a benefit. The benefits are also quite small in ruptured membranes or preterm labour, with an NNT of about 67, and that should be taken into account in the decision making.
Nevertheless, there has been widespread uptake of ANS prior to late preterm delivery, hopefully there will be good long term follow up of the babies in ALPS, that is the only way we will tell in the future whether this major change in practice is actually improving outcomes for babies, or not.









Thank you, for yet another excellent post.
Denmark has used ANS for GA < 34 since the late seventies, but adaption for planned c-section GA 34 to 36 is controversial and we don’t use it for pregnancies at risk. A primary concern is for the well known neurotoxicity of glucocorticoids. Late preterms are presumably at less risk for such effects, but I do not think we have data to say that the risk is zero. I would suggest that it is problematic to design and execute trials of ANS, without powering for and planning for neurocognitive follow up of infants.
Further, none of the original AS trials, that contributed data to Crowleys meta analysis (https://pubmed.ncbi.nlm.nih.gov/7631713/), did long term follow up. In addition all of these trials only analysed outcomes for children whom were actually born preterm, and none of them analysed outcomes for foetuses exposed to ANS but born at later gestational ages. Recent observational studies have indicated ANS exposure may have adverse effects for foetuses who progress to be born at term (https://pubmed.ncbi.nlm.nih.gov/32427304/).
Treatment and prognosis for infants born in the late weeks of todays ANS treatment spectrum has improved dramatically since Liggins did his RCT. Therefore, it could be suggested that todays most pressing question isn’t wether GA 34 to 36 should be offered ANS. Rather, we should perhaps aim to test whether foetuses GA 32 to 33 at risk of preterm birth overall benefit from exposure to ANS? Needless to say, such trials would be costly, as they would have to be very large and follow up should continue well into school age.
The other important issue, that Alan Jobe has been emphasizing for a long time, is that the effective dose of betamethasone is unknown. We are still using the same dose as Liggins and Howie, which was extrapolated from sheep data, and there has never been an adequate dose-ranging trial. I think it would be entirely ethical to randomly compare a standard dose to a reduced dose (which seems very effective in sheep studies), and such a study which would focus on short term efficacy, should also examine long term safety. From simple principles, if a lower dose is as effective, then there is no way it could be more harmful. I found Gyamfi-Bannerman’s dismissal of the potential harms “Gyamfi-Bannerman C, et al. Concerns regarding the use of antenatal betamethasone in late preterm gestation: is the evidence of potential harm being overstated? BJOG. 2016;125(8):923-4” unconvincing. The observational data of sibling pairs from Raikkonen that you refer to is rather concerning, I discussed it in the blog when it was published, and it certainly suggests that we need to find ways to 1. better target mothers who will actually deliver preterm, 2. define a lower dose which is effective and 3. have long term outcome data which shows that we are really benefiting the babies. A 3% reduction in respiratory morbidity, which is a transient short term benefit, would be difficult to justify if there is any indication of long term harm. We just don’t have good enough data to say that use in the late preterm is safe.
Thanks again for enlightening us.
I guess this paper that popped up on my alert list this weeks adds further to the discussion regarding pro/cons for late preterms and ANS:
https://pubmed.ncbi.nlm.nih.gov/35404395/