I have been continuing to think about this issue, so will subject you all to some of my thoughts. The large RCTs of medication for closure of the PDA show no benefit, and some evidence of harms, but we all know that the PDA usually closes shortly after birth in the term infant, and there is a statistical association between a persistently patent ductus in the very preterm infant and several neonatal complications.
So why don’t babies benefit from closure of the PDA?
I think there are 2 possibilities. One is that the medications are the problem. They are not very effective, and we expose the babies to reduced urine output, and other side effects, for unreliable constriction of the PDA, and a relatively small proportion of complete closure of the duct. Especially among the most immature babies, the drugs don’t work very well, and they have more secondary effects. Perhaps there are benefits to closing even modestly sized PDAs, but they are overwhelmed by the adverse effects of the medications we are using.
It is interesting that in the causes of mortality, as far as they can ever be definitively determined, there does not appear to be a difference in NEC, according to the meta-analysis that I referenced before, despite concerns of some that there may be an impact on GI perfusion of the meds. That SR/MA does, however, show an increase in severe IVH, PVL, and culture positive sepsis. None of which are individually statistically significant, but all of which suggest potential impacts that we often don’t consider. The decrease in urine output could also lead to an increase in lung water, ventilatory requirements and lung injury. So maybe the medications are the problem.
The other possibility is that the PDA is not etiologically linked to the associated complications, but is a bystander. I think for PDAs with a small shunt, this is probably the case. Centres with a very restricted approach to the PDA do not, generally, have worse outcomes overall. However, I think that is likely that there are babies who benefit from closure, including, perhaps, early closure.
Are there any babies, in controlled trials, who seem to benefit from PDA closure, in the current literature? Afif El-Khuffash and colleagues performed a secondary analysis of their trial which only included infants with a large shunt. The original trial publication (El-Khuffash A, et al. A Pilot Randomized Controlled Trial of Early Targeted Patent Ductus Arteriosus Treatment Using a Risk Based Severity Score (The PDA RCT). J Pediatr. 2021;229:127–33) showed a null result, but it was intended as a pilot trial, and was therefore under-powered for clinically important outcomes. The secondary analysis, was, therefore, even more underpowered, (Bussmann N, et al. Patent ductus arteriosus shunt elimination results in a reduction in adverse outcomes: a post hoc analysis of the PDA RCT cohort. J Perinatol. 2021;41(5):1134–41) there were only 17 infants who had successful closure of the PDA in the group with large PDA shunts who were randomized to treatment with ibuprofen. Among those who actually managed to close their PDA, they were better off than the placebo group infants in terms of “death or BPD”, 5/18, compared to 18/30 control group babies, which isn’t “statistically significant” by a simple chi-square. All secondary analyses are suspect, but, it does suggest that there may be a subgroup of babies who benefit from PDA closure. Interestingly, the active treatment group infants who did not close their PDA, were actually worse off (11/13 had “death or BPD), and possibly had more deaths.
One way of eliminating the adverse medication effects would be to close the PDA without medication. For those who are not aware, there is a trial in progress of ductal closure by catheter, the PIVOTAL trial, which randomizes infants of 7 to 32 days of age, <28 weeks GA, and over 700 g birth weight, who have a PDA score of 6 or more. The PDA score in use for this trial has been adjusted from that PDA RCT. I asked my good friend, the PI of the study, Carl Backes which score they were using and he very helpfully sent me this table, which I am sure he won’t mind me sharing.
| ECHO-Based Scoring System | 0 points | 1 point | 2 points |
| PDA size indexed to weight | <1.5 | 1.5-3 | >3 |
| Mitral inflow velocity (E) (cm/s) | <45 | 45-80 | >80 |
| IVRT (ms) | >50 | 30-50 | <30 |
| PV D-wave (cm/s) | <0.3 | 0.3-0.5 | >0.5 |
| LA:Ao | <1.3 | 1.3-2.2 | >2.2 |
| LVO:RVO ratio OR LV-VTI:RV-VTI* | <1.5 | 1.5-2.0 | >2.5 |
| Descending aorta AND/ORceliac AND/OR middle cerebral artery flow | Forward | Reversed | |
| PDA size indexed to weight= (minimum PDA diameter [mm]) / (weight [kg] at time of ECHO); IVRT=isovolumic relaxation time; PV=pulmonary vein; LA:Ao=left atrium to aortic ratio; LVO=left ventricular output; LV-VTI: LV output velocity time integral; RVO=right ventricular output; RV-VTI: RV output velocity time integral*obtain both LVO:RVO and LV-VTI: RV-VTI measurements; the highest score from either ECHO parameter is counted towards overall score*use the high suprasternal arch view to obtain the post-ductal diastolic flow measurement; reversal present from an abdominal view is acceptable, but the “absence of reversed flow” from an abdominal view may be a false negative, thus the need for the suprasternal view. | |||
I hope that the PIVOTAL trial is planning a subgroup analysis by PDA shunt size or by gradations of the score, I think it is unlikely that a score of 6 or more was exactly the right choice, as the threshold for enrolment, but I could be proved wrong. The planned sample size was 240, and they are apparently about 75% complete. The trial will at least eliminate the problem of medication toxicity, but instead introduces the risks of catheterisation!
Catheter closure has rapidly been growing in the USA, the latest data I can find are from 2021, and they show things like the following, from the NICHD NRN
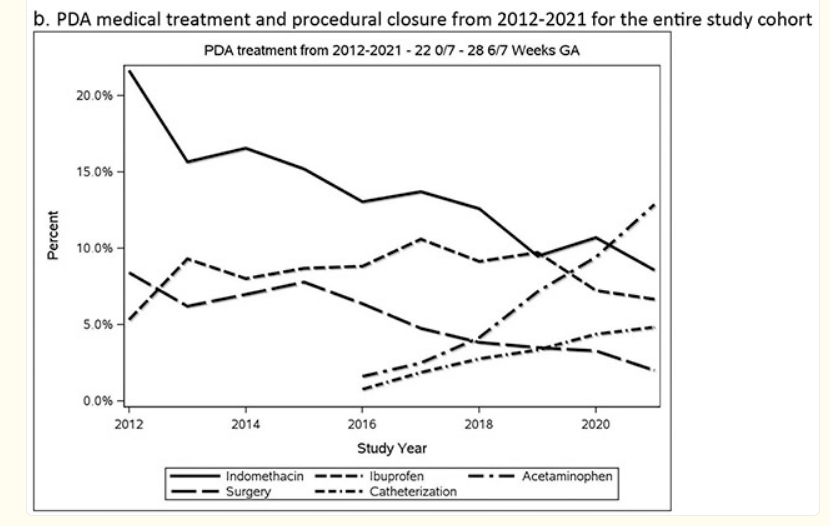
Indomethacin is disappearing, ibuprofen is relatively stable, acetaminophen has become the commonest intervention for the PDA, and the progressive disappearance of surgery is being replaced with the very rapid growth of catheter closure, being nearly 5% of babies born <29 weeks.
Given that there is an absolute lack of evidence that catheter closure of the PDA improves any clinical outcomes, it is very depressing to see that 1 in 20 extremely preterm infants in the NRN are being subjected to this procedure, almost all of them outside of any clinical trial.
Equally depressing the “Society for Cardiovascular Angiography & Interventions” (SCAI, a new one for me) have just published a position statement with a pre- and post- procedure checklist. Carl Backes, and his co-author wrote an accompanying editorial and they were much kinder about this position statement than I would have been. My response would have been : WTF? How can you write a position statement for a procedure completely lacking in supporting evidence that it is effective or safe, in terms of clinically important outcomes? Surely the position statement should have stated, that this procedure should not be performed outside of a prospective study.
Hopefully the PIVOTAL trial will prove to be pivotal, and will have a clear result, of benefit or no benefit. The primary outcome is ventilator free days, a reasonable way of analyzing the impacts on ventilator dependence or death (as deaths will be counted as 0 VFDs). Although I would have wanted other outcomes, in particular some evidence that effects on lung injury, using measures that are important to families, are improved, without an increase in mortality.









Keith: I appreciate your making sense for a rational approeach to the PDA. In 1981 J Pediatr we published an underpowered RCT regarding early closure. 50 years later Mikko Hallman and the Finnish group published a similar study with les toxic acetaminophen a much less toxic agent to affect closure. D.K. Edwards published the radiographic effects of closure and the diminished time on assistedd ventilation at the time (IPV via ETT). I recall only about 35% of infants ≤ 1500 grams met a less sophisciated ECHO criteria for a HS PDA suggesting that 63% did not. At about the same time Stahlman’s group reported on outcomes of infants who after 30 days had their PDA closed surgically with not great outcome.
The rush to constrict the PDA (championed by L. Gluck) was ill-advised for infants and unpublished study when he was at UC Irivine found an increased mortality. I find the “rush” to plug the PDA unwarranted and just because it can be done should not be translated that it should be done. Allen Merritt
5
. 1981 Aug;99(2):281-6.
doi: 10.1016/s0022-3476(81)80479-9.Early closure of the patent ductus arteriosus in very low-birth-weight infants: a controlled trial
T A Merritt, J P Harris, K Roghmann, B Wood, V Campanella, C Alexson, J Manning, D L Shapiro
DOI: 10.1016/s0022-3476(81)80479-9
PMID: 7019406