There are 2 main causes of intestinal perforation in the very preterm, Necrotising Enterocolitis and Spontaneous Perforation. NEC, as I have previously discussed, may be a convenient name for a few different conditions which present in a similar fashion. In particular NEC in infants with congenital heart disease, in infants with Hirschsprung disease, and in infants with gastroschisis, may all have differing pathophysiology, and methods to prevent and treat them may also differ. For the moment, very preterm infants without those conditions who develop NEC are considered to have a single disease, but it may be that more than one pathophysiologic process can lead to what we call NEC. After we introduced probiotics in our nursery the incidence of NEC fell, but also the average age of the infants who presented with NEC was substantially younger, making me wonder if the balance of different aetiologic factors is different in younger versus older cases.
Most database publications have noted that NEC overall is quite a lot more frequent than Spontaneous Intestinal Perforation (SIP) and even the NEC infants requiring surgery (mostly for perforation, but not all) are a larger group than SIP.
The Canadian Neonatal Network, for example, reported that among 2,019 infants <28 weeks gestation, 39 (1.9%) had spontaneous intestinal perforation, 61 (3%) had perforated necrotizing enterocolitis, and 115 (5.7%) had non-perforated necrotizing enterocolitis.
The German Neonatal Network reported, among 8,022 babies under 1500g birth weight (and <37 weeks gestation), 177 cases of surgical NEC and 123 with SIP.
Which makes the distribution of cases in this newly published trial somewhat surprising, with rather more SIP than surgical NEC. Blakely ML, et al. Initial Laparotomy Versus Peritoneal Drainage in Extremely Low Birthweight Infants With Surgical Necrotizing Enterocolitis or Isolated Intestinal Perforation: A Multicenter Randomized Clinical Trial. Ann Surg. 2021;274(4):e370-e80.
In this trial, babies of <1001 g birth weight, and less than 8 weeks postnatal age with either NEC or SIP, for whom a decision to perform surgery had been made, were randomized. They either were directed towards initial laparotomy or initial peritoneal drainage in the NICU under local anaesthesia. Planned sample size was 150 per group in order to have an 80% power of detecting a 15% absolute improvement in the primary outcome, which unfortunately was “death or NDI”. For this trial “NDI” was cerebral palsy with a GMFCS of 2 or more, a Bayley III cognitive composite score of <85, or blindness or deafness, at 18 to 22 months corrected age.
This trial is a perfect example of why we should abandon this outcome. Death is common in this group of babies, neurodevelopmental problems are also common among survivors. But this definition of NDI, which is given the same weight in the outcome as being dead, includes infants who will mostly be ambulant but walk with some difficulty, i.e. they will limp. A Bayley cognitive score of <85 has very little correlation with functional capacities or outcomes. Ask a parent of a deaf or blind child if that outcome is equivalent to them being dead; or ask them themselves when they are old enough! There is also no good reason for supposing that mortality and neurological impairment or developmental delay will change in the same direction with these interventions. This would have been a perfect trial for a “win-ratio” approach, with a substantial risk of death, and a major risk of long term difficulties among survivors. What I want to know, in order to make the best decision for my patients, is whether mortality will be affected, and if not then is the long term outcome better or worse? If mortality is better with one approach rather than another, are all the extra survivors profoundly impaired? (That would be a unique result in neonatology if it happened, but I guess it is possible). Becaue if not, then mortlaity trumps all the other outcomes.
The authors achieved a very high follow up of 96%, but the difficulties on doing such a trial are illustrated by the fact that enrollment took 10 years!
They did indeed finally enrol 300 babies, but as mentioned, somewhat surprisingly, there were 95 with NEC and 213 with SIP. This immediately makes me concerned about some sort of selection bias in the enrolment process. It may be that infants with what was thought to be definite NEC were less likely to be approached for consent, I would guess that for many of those babies the care team thought that going straight to laparotomy was appropriate, but potential randomisation to bedside drainage was not. Among the NEC babies enrolled nearly half were on vasopressors at enrollment, suggesting that babies who were more unstable, and therefore thought potentially worth consideration of peritoneal drainage, were more likely to be enrolled.
With those provisos, lets look at the results. In a strictly trial design sense, there was a nul result. No difference between the 2 approaches for the primary outcome, 69% death or NDI with laparotomy, and 70% with peritoneal drainage. About half of the drainage babies went on to have a later laparotomy, about 1 in 5 of the laparotomy babies needed another laparotomy later on. Mortality for the 2 randomized groups was similar, 29% vs 30%.
Mortality, however, was quite different among those babies who the Surgeon thought, pre-op, were NEC rather than SIP, (there were 7% of the laparotomy babies who actually had neither NEC nor SIP, but another diagnosis including volvulus and gastric perforation). Among the “thought to be NEC” babies, mortality was 40% with laparotomy and 51% with initial drainage, the “thought to be SIP” babies mortalities were 23% (laparotomy) vs 19% (drainage).
Although neither of these differences is “statistically significant”, the authors performed a Bayesian analysis of the results (Jon Tyson is the second author!), which showed that initial laparotomy is much more likely to lead to a reduction in “death or NDI” in the NEC babies with a probability of 97%, and is somewhat less likely to be beneficial in the SIP babies, only 18% chance of being better, compared to 82% chance of being worse.
I think these Bayesian analyses are a reasonable attempt to explain the implications of the data, when, despite the enormous efforts involved in a trial lasting 10 years, final sample sizes are modest. Obviously not as good as having a trial with 1000 babies per group, but it does suggest that such a large trial would be very unlikely to show a benefit of intial drainage among babies with surgical NEC.
Among babies with a pre-op diagnosis of NEC, there were 25 long-term survivors after laparotomy (of 42 infants), and 26 survivors of the 52 who had initial drainage, of whom 25 were followed up. Of the laparotomy babies, 13 of the 25 survivors had no “NDI”; among the drainage babies 9 of the 25 had no “NDI”. The fairly small numbers lead to low confidence in the results, but all the differences in long term outcomes in these babies favour intial laparotomy. Among the SIP babies there are fewer babies with CP (GMFCS 2 or more) after initial laparotomy, but somewhat more babies with lowish Bayley scores. Here are the details of the “NDI” outcome, from the supplemental materials:
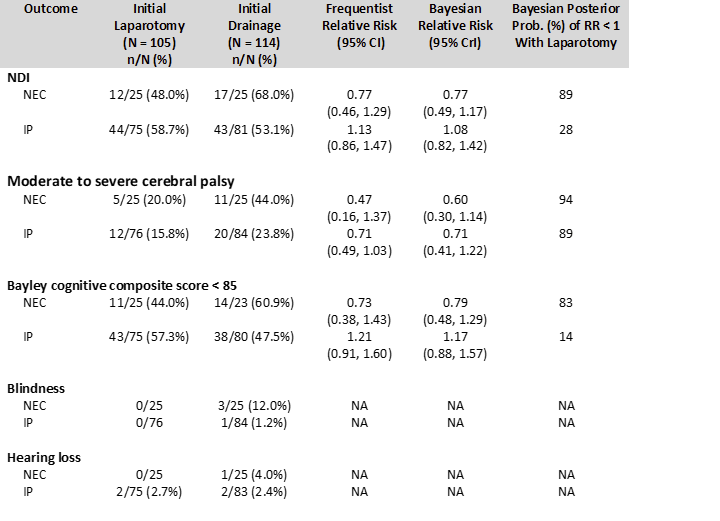
As an article I just published with Elliott Weiss and Stephanie Kukora (Weiss EM, et al. Use of composite NICU research outcomes for goals of care counselling creates ethical challenges. Acta Paediatr. 2021) discusses, not only are such composite outcomes a problem for clinical trial design and interpretation, they make counselling of families more difficult, what are we to say to families when trying to decide which approach to take for an infant with NEC who needs surgery? This trial shows, strictly, no difference between the two approaches. But I think it is much more helpful to parents to be able to say, for a baby with a diagnosis of NEC and an indication for surgery, that mortality is lower with immediate laparotomy, and in the long term, despite significant risks with either approach, outcomes seem to better with immediate surgery (48% “NDI” among survivors with laparotomy and 68% “NDI” among survivors after initial drainage, in this trial).
There are 2 previous trials with similarities to this one. A European trial of 69 babies with perforation, including NEC and SIP babies (Rees CM, et al. Peritoneal drainage or laparotomy for neonatal bowel perforation? A randomized controlled trial. Ann Surg. 2008;248(1):44-51) showed somewhat lower mortality with laparotomy, 33% vs 40%. In that trial over 60% of the babies had NEC confirmed if they had surgery. A previous North American trial of 117 infants (Moss RL, et al. Laparotomy versus peritoneal drainage for necrotizing enterocolitis and perforation. NEJM 2006;354(21):2225-34), was supposedly limited to infants with NEC, but the entry criteria as described in the article would have included SIP “preterm infants (birth weight, <1500 g; gestational age, <34 weeks) with evidence of intestinal perforation, including free intraperitoneal air on an abdominal radiograph (96 infants); stool, bile, or pus found at paracentesis; or clinical evidence of perforation in the joint opinion of the attending surgeon and the neonatologist. This definition included both infants with extensive disease and others with focal perforation”. 60% of their subjects did not have pneumatosis on abdominal x-ray. In that study mortality was 35% in each group.
If I try and put all of this together, it looks to me that infants with perforated NEC should probably be directed, if possible, to immediate laparotomy. There is no advantage to initial drainage, and some evidence of lower mortality, and perhaps better long term neurologic and developmental outcomes, with immediate laparotomy. Also, 7% of the infants may turn out to have another diagnosis, which probably mostly require surgery also.
In infants with probable SIP the question is more difficult, but there doesn’t appear to be a clear advantage to delaying laparotomy, there may be a slightly higher survival with initial drainage, but in the long term outcomes seem rather balanced; with the highly problematic outcome, of uncertain real importance, that a cognitive Bayley composite <85 was a little more frequent after laparotomy than after drainage.
Makes me wonder, yet again, how we got to the point where a Bayley cognitive composite of 86 is fine, but a Bayley cognitive composite score of 84 is a disaster, an impairment, equivalent to death! Dichotomizing human development into normal and subnormal is an affront to me, and I think to most of those who actually do follow up.
This review points out a number of problems with NEC research, one is the lack of good definitions. A recent publication by Janet Berrington and Nick Embleton (Berrington J, Embleton ND. Discriminating necrotising enterocolitis and focal intestinal perforation. Arch Dis Child Fetal Neonatal Ed. 2021) has illustrated some of the difficulties: reviewing a large cohort, many cases that had been classified as NEC were found after extensive review to more likely be SIP. Some of the babies satisfied published diagnostic criteria for one disease, but after review, including surgical histopathology, they had the other. I am rather sceptical in general about the usefulness of many published biomarkers, which often seem to be a way of increasing numbers of publications rather than helping clinical practice, but here is one situation where a good discriminating biomarker might really help to differentiate these conditions, and also point, perhaps to other diagnoses. This study shows clearly, yet again, that CRP is of no use (not always being elevated with either diagnosis), and most of the articles that I have looked at don’t seem to have tried to differentiate NEC and SIP using their biomarker.
Surgical NEC has a very high mortality in all of these studies, and the mortality of SIP is not negligible. Neurological impairments and developmental delay are frequent with surgical NEC, medical NEC, and probably following SIP also. The fight to reduce these diseases has to intensify, we have made relatively little progress in recent years. Mother’s milk, and donated human milk when mother’s milk is not available are interventions that reduce NEC compared to formula feeding, but we have known that for years. Finding ways to normalise the intestinal microbiome are are potential part of the solution, but administration of current probiotics is only partly successful, both in normalising the microbiome and in reducing NEC. Avoiding antibiotics whenever you can, and having a feeding protocol are both helpful, but neither can eliminate NEC, and probably have no impact on SIP.
I have seen too many babies die of NEC in my career, I sincerely hope that, during the careers of my trainees, we will be able to stop these scourges.








