This was an idea I had not heard about prior to seeing this newly published trial (Mank E, et al. Efficacy and Safety of Enteral Recombinant Human Insulin in Preterm Infants: A Randomized Clinical Trial. JAMA Pediatr. 2022). The introduction is very limited on the prior justification for the trial, but the beginning of the discussion gives a lot more background.
The discussion section notes that, in previous animal studies “the small intestinal weight and intestinal disaccharidase activity (ie, lactase, sucrase, maltase) were significantly higher in both piglets and rats treated with either enteral rh (recombinant human) insulin or enteral recombinant porcine insulin relative to controls, suggesting that insulin has a key role in promoting intestinal maturation. The effect of enteral insulin on the intestine seems to be mediated by insulin receptors, which have been observed on both the apical and basolateral enterocyte membrane of various animals” and apparently insulin receptors have been seen in human foetuses up to 19 weeks, but only on the basolateral membrane of the enterocytes, not apically. There is apparently also quite a lot of insulin in human milk in the first few days after delivery, and an interesting prior study from Deborah O’Connor’s group in Toronto showed that standard Holder pasteurization decreases human milk insulin concentrations by about half.
They therefore performed this study to see if adding a new powdered insulin preparation to human milk feeds would improve GI function, as measured by time to tolerating full enteral feeds, among preterm infants <32 weeks gestation.
The first disappointment I had with this trial was that the investigators excluded infants less than 26 weeks gestation. I can’t see any good reason why more immature babies should have been excluded from the trial, more immature babies are more likely to have feeding intolerance, and including higher risk patients improves study power. Also, if it works overall, then surely you would want some evidence that it works in the highest risk patients. Excluding higher risk, more immature, patients from trials means that we cannot develop the evidence base needed to improve their care (Barrington KJ. The most immature infants: Is evidence-based practice possible? Semin Perinatol. 2021:151543). There were several exclusion criteria, including infants significantly SGA, needing more than 60% oxygen, having a blood count suggestive of infection, etc. Babies were eligible within the first 5 days of life if they had started enteral feeds. Babies were randomized to receive either 400 micro-units of insulin per mL of milk, or 2000 micro-units/mL or placebo, in a masked fashion.
The next disappointment is that most of the attached protocol is redacted. Which is truly weird. The study procedures part of the protocol from section 9.1, screening and eligibility procedures to 9.5 discontinuation/withdrawal of infants are blacked out, the 10 pages with the important details of the study protocol look like something from Trump’s White House or a report into parties at 10 Downing Street. I wanted to read the protocol because I wasn’t clear about one part of the published article, where it states that, in babies receiving exclusively maternal milk, treatment was not initiated until 72 hours post-partum. It wasn’t clear to me what that meant for the other infants, and what would happen if the mother wanted to breast feed, but baby received partially banked human milk, which is why I wanted to read the protocol… I have no idea what they are trying to hide.
I think the delay in maternal breast milk fed babies is because there is a lot of insulin in human milk in the first 3 days, so they wanted to avoid an overdose in those babies. Presumably the intervention was started immediately after randomization in the other babies.
The primary outcome of the trial was the time to full enteral feeds, defined reasonably as 3 consecutive days of at least 150 mL/kg/d of milk. The sample size was calculated based on a reduction from 8 days to 6.6 days, as shown in a pilot study. I find that outcome a bit problematic, it isn’t a terrible idea, but really, who cares? I think the reason for caring about time to full feeds is that more prolonged feeding intolerance is associated with increased other complications, such as late-onset sepsis, cholestasis and increased costs. 1.5 days less of partial parenteral nutrition is really only of clinical significance if it is, firstly, clearly very safe, and secondly leads to reduced other complications. I understand that powering a trial for those other complications is going to lead to very much larger sample sizes, but that is also the only way to be sure that the intervention is safe. If even a small proportion of babies have an adverse secondary effect of enteral insulin, then a reduction of 1.5 days of time to full feeds becomes irrelevant. I think at this stage of investigation, time to full feeds was a reasonable compromise, but doesn’t give me a strong reason to consider introducing a new intervention.
The sample size calculation arrived at 150 babies per group, in a multicentre design, with 2 gestational age strata. After 225 babies were randomized, an interim analysis showed that the calculated power for finding a reduction of 1.5 days in time to full feeds was <35%, so the DSMC proposed stopping the trial. By the time the trial was actually stopped 303 babies had been randomized. In another move which is not fully explained, babies who were in the trial, but had not yet reached full feeds had the intervention stopped, in addition, there were some babies who had the intervention stopped because they were transferred to non-study centres. As a result there are only data about the primary outcome for 261 infants.
Although the trial was stopped early for futility, the final results actually show what is potentially a valuable reduction in time to full feeds in the insulin groups. Median time to full feeds was 10 days in each of the insulin groups compared to 14 days in the controls. Which was twice as great as the hypothesized difference. The difference looks unlikely to be due to random effects, the 95% compatibility intervals were 1 to 8 days shorter time to full feeds. Other outcomes were not much different between groups, if you put the 2 insulin groups together, mortality was about the same 3% insulin vs 4% control, late-onset sepsis was slightly lower with insulin 12% vs 15% and NEC was lower, a difference which, if confirmed in future larger trials, might be a clinically important difference, 5% vs 10%. My back of the envelope calculation shows that difference to be compatible with no real impact of insulin on NEC, or, obviously, with a major reduction.
Early stopping of trials for futility is a common occurrence which seems to be getting more frequent. There are scientific and ethical reasons for questioning the practice, which are illustrated by this trial. Scientifically speaking, the risk of stopping early for predicted probable futility is exactly what this trial has shown, that the trial, when fully analyzed, may show a real difference between treatments, even a difference greater than hypothesized, but being stopped early, of course, reduces the power of the trial. It is easy to imagine situations in which a similar trial may be stopped and show a difference which is not quite convincing, and reported as “not statistically significant” but if it had been continued, and accrued the expected number of participants would have been a positive trial.
Other trials have had similar occurrences, the following figure, for example, is from a cancer trial (for which futility analyses are common) The calculated power of the trial to produce a significant result is plotted over time, which shows that it varied substantially as the results accumulated, the point at which the power dropped was when the pre-planned futility analysis was performed which led to stopping the trial. The lower graph shows the actual primary outcome (for PFS, progression free survival) at the end of data collection, which was better with the new regimen than the old one.
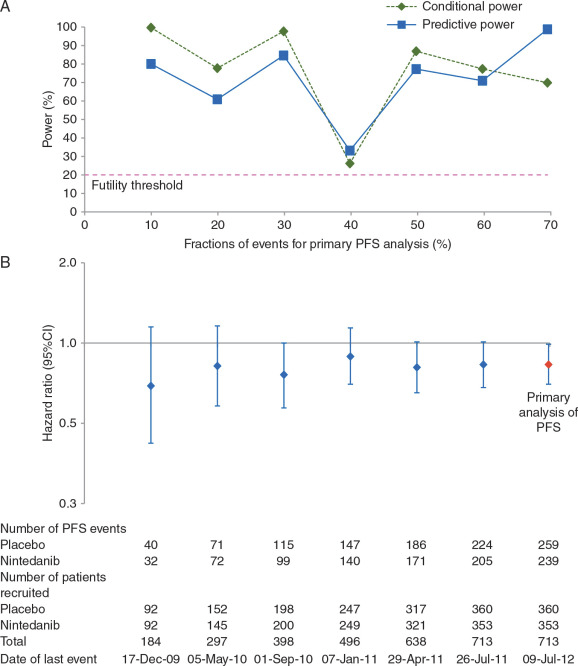
In our current trial there were no safety issues identified at all, no evidence of harm. Also it is hard to believe that oral insulin powder is a hugely expensive intervention, which might be an issue with continuing some cancer trials, especially industry sponsored trials where the medication might be extremely costly and potentially toxic.
Ongoing monitoring of trial outcomes to reduce risks of the trial to participants is very important. But where there are no identified risks of the intervention, and where being in the trial does not prevent the participant from receiving other interventions, (I can imagine in cancer trials that some of the considerations are different), then the threshold for stopping a trial should be much higher.
From an ethical point of view, I think we have a responsibility to families that participate to ensure the trial is as useful as possible to the community. To expose families to the stress and potential risks of an intervention, and then stop a trial before you have any reliable answers is very questionable. In particular, I think it was wrong in this trial to stop the intervention in babies who had already been randomized and started the intervention. Those families had already consented to be part of the trial, and had started the intervention (insulin or placebo) but then had the intervention stopped part way through, meaning they were exposed to the stress and to the risks of an RCT but were not permitted to contribute to the final results. Surely, once babies are randomized they should have continued in their randomized group until they reached primary outcome criteria, and for the 28 day intervention period.
I think if the DSMC had found increased risks for the insulin group, that would have been an entirely different situation. Also, if the primary outcome results were clearly going in the wrong direction, with an increase, rather than the expected decrease, in time to full feeds with the insulin, then I think the decision to stop for futility would make more sense. Instead we are left with a trial which did not reach planned sample size, in which 42 of the 303 enrolled babies did not even contribute to the primary outcome. A trial which is, therefore, only suggestive of a benefit.
Imagine if the trial had continued to the planned sample size, it is quite possible that an important reduction in NEC might have been found, as well as a potential reduction in late-onset sepsis. At least we would have had more reliable data to proceed with more investigations.
The next disappointment is that the study finished in 2018, and only short term outcomes are reported. Why did it take 4 years to get this in press? It seems an unnecessarily long delay.
The final disappointment is that many of the babies did not receive milk from optimal sources. Although the study started in 2016, and human milk banks were not as widely available as they are now, there were several babies who received formula alone, and many who received a combination of milk sources; the “mixed” category in the results includes babies who received both maternal and banked milk, as well as those who received both human and artificial milk, and it is not clear how many received artificial milk. As the standard of care is now maternal milk feeding, and banked human donor milk when sufficient maternal milk is not available, for preterm babies at risk of NEC (such as all the babies in this study) it is important to know if these potential advantages of enteral insulin apply to babies who received only human milk.
The potential advantages of adding insulin to the feeds of very preterm babies suggested by this study are such that further trials are definitely worthwhile. Those trials should only study babies receiving milk from optimal milk sources, they should include the higher risk babies (less than 26 weeks, small for gestational age, abnormal antenatal dopplers) and should have much stricter rules for terminating the trial for futility. They should be powered to investigate clinically important outcomes such as late-onset sepsis and NEC.









Dear Keith,
Nice to hear from you and thanks for picking up this trial and making the results distributed further. Yes, in every trial there is room for improvement and certainly a multinational/multicenter trial can have challenges. But I am not at all disappointed as I think this trial may help in further improvements for these vulnerable infants. Yes, we do need more studies, but one well designed large one could do the trick and the good news is that this is planned.
That trial will include smaller infants as well, but including the most vulnerable ones (from 23 weeks onwards) may alos pose it’s threats to a study. Mortality rates are much higher in those infants unfortunately, with concomittant more issues than we already had on DSM aspects. That it took a while before we were able to publsh these data is also due to the much longer time it took at several journals we approached for the peer review process in the last year. COVID 19 caused many problems, both from ouw own side but also from reviewer perpectives and did not increase the speed.
But overall we are happily surprised on the effect, it may help in reducing time on parenteral nutrition, on sepsis and hopefully as well on NEC incidence. Lets wait for those results.
Best wishes Hans
Thanks Hans, for the comment, and I am really pleased that another larger trial is planned. I think the results of this one are promising enough that we really need that. I agree that it is problematic to include the most immature babies, but I think its more problematic to exclude them; especially as many centres now routinely offer active care at 23 and 22 weeks gestation. We desperately need a good evidence base for their treatment. Those babies are so physiologically immature that they may not have the same response as more mature infants, and a stratum of babies under 25 weeks would be great.
As for my other comments, I think I overdid the “disappointments”, the most disappointing thing is people who don’t do the research, and introduce new interventions without good evidence! In contrast, your group has investigated something novel and potentially important, with a masked RCT. I would guess that the early stopping of the trial, which ended up actually being positive, was a disappointment to the investigators as well!
It would be great to know the impact of the enteral insulin among babies who only received human milk, but, as you have shown, there isn’t much insulin after the first few days, and there probably isn’t much in most donated milk, especially after Holder pasteurisation; it might be just as effective for the human milk fed babies as formula-fed.
Dear Dr. Barrington, I always enjoyed your reviews. There is a minor correction – the dose is 400 or 2000 micro IU of insulin per ml of breast milk (not 400 or 2000 units/ml as the review mentioned).
Thank you for spotting that, I have corrected the post.