The standard method of pasteurization of donor breast milk, and I believe the only method approved by HMBANA (the human milk banking association of north america), is similar to what Louis Pasteur himself came up with a couple of centuries ago (in about 1865). I’m not sure why its called Holder pasteurization, it may be because the temperature is “held” at 62.5 degrees for 30 minutes, or maybe it’s named after someone. In any case, it is effective in killing most bacteria, not including spore-bearing forms (like Bacillus cereus), but, unfortunately, inactivates white cells, kills probiotic organisms and denatures many large molecules, especially proteins.
Some of the advantages of human milk are therefore impacted by Holder pasteurization (HP), and there are a few review articles available (including this one from the European MBA) which discuss the available pasteurization methods. Other means of pasteurization that have been evaluated, include ultraviolet treatment, high hydrostatic pressure, and most particularly High Temperature Short Time (HTST) which uses temperatures of about 72 degrees for 15 seconds. All the alternative methods seem to be equally good at inactivating bacteria, and many viruses, but the alternatives have much less negative impact on the other beneficial components of milk (Oligosaccharides are relatively unaffected by HP). The major current problem with changing over to alternatives are the new equipment that will be required, and the lack of good evidence that it actually makes a clinical difference.
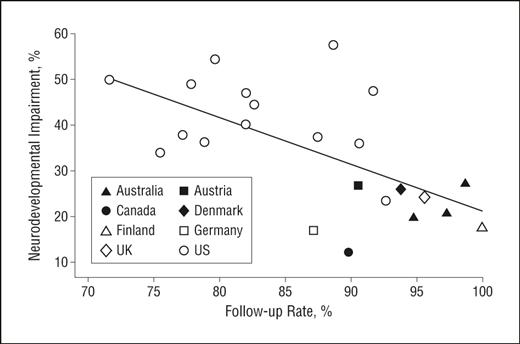
A recent RCT from 2 NICUs in Madrid compared supplementing mothers milk with pasteurized donor milk that used one of 2 different pasteurization methods, HP and HTST. They enrolled 213 and analyzed results from 160 ELBW infants, with the primary outcome being the occurrence of CLABSI, or a positive blood culture in an infant who had a central line in place for more than 48 hours, and in whom the culture was drawn during catheter use or within 48 hours after.
The most striking result is the enormously high frequency of CLABSI in those units, in both groups, 42% with the HTST and 46% in the HP group. There was no statistical difference between groups, but the study was designed to have 150 babies per group, and powered for a 33% reduction in the outcome. They stopped adding patients after 160 were actually enrolled, which is never really explained, the confidence intervals for the relative risk of catheter related sepsis were 0.68-1.26 with HTST compared to HP, so a large potential benefit is consistent with this null result.
The definition of late-onset sepsis is quite rigorous, and required a positive blood culture, after 72 hours of life, with a central catheter in place (or removed <48 h) and at least 2 of a sequence of clinical signs, or in the case of CoNS also other lab results (CBC results or CrP raised). When expressed per 1000 catheter days, the incidence in the two groups was about 18/1000 catheter days.
As a comparison, among babies <1kg birth weight in the CNN, all LOS combined (which is any positive blood culture, not requiring any other criteria, either with or without a central catheter in place) was 28%. In a publication from the German Neonatal Network, they showed “only 45% of primary BSI were CVC related in the <1000 g birthweight group” in other words, only about half of LOS was catheter related. That percentage is going to vary a great deal, depending on how central catheters are used, and the GNN also showed also a large variation in use. Nevertheless, the infections reported by this new Spanish study are only a subgroup of all the LOS in their infants; again in comparison with the Canadian Network, among infants <33 weeks the incidence of CLABSI was about 7.5/1000 catheter days.
As far as I can tell, the Spanish study did not enrol particularly high-risk babies, some were eliminated because of a “risk of early death”, they seem to be a group with relatively standard risk profile.
I think that different ways to try and preserve the advantages of human milk, by using different pasteurization methods is really important. However, by themselves, such improvements will not be likely to have a huge impact on LOS rates, especially when only a small proportion of the milk received is the donor milk. Enteral colonization with pathogenic, mostly Gram negative, organisms is a known common occurrence prior to blood stream infection. This recent publication for example, Schwartz DJ, et al. Gut pathogen colonization precedes bloodstream infection in the neonatal intensive care unit. Sci Transl Med. 2023;15(694):eadg5562, showed that GI colonization with the same organisms was very common, and that the pathogens were more abundant, in babies who later developed LOS. The anti-infective properties of milk will have an impact on gut colonization, and it is likely that different pasteurization methods will have an impact on those benefits.
There are many other things we do that can affect the incidence of LOS, I note that in the Spanish trial the use of “anti-acid” medications was very common at 20 to 30%, there are no details, and they may have been given after the episodes of sepsis, but I think there is very little indication for the use of such medications, which clearly can increase gut colonization by reducing one of the important barriers.
I just hope this new study doesn’t discourage the on-going development of better ways to pasteurize donor human milk which preserve more of the advantages. I think we need more, adequately powered studies to, firstly, show impacts on the intestinal microbiome of different pasteurization methods, and, if possible, following that, the clinical benefits. But we must take into account the large number of other risk factors for LOS, and reinforce quality control. Stringent quality improvement methods can reduce LOS in the very preterm, and have to be a priority wherever ELBW infants are cared for.