As usual, the annual meeting of PAS had too many things going on simultaneously to be able to get to all the interesting looking neonatal research. But here are a few things, that were of interest to me, and which I either got to in person or wanted to.
How much CPAP after extubation?
The Éclat trial (happy to see a French trial acronym) studied babies <28 weeks who had received surfactant and were on caffeine and were about to be extubated. They were randomized to either standard CPAP pressures after their first extubation (6-8 cmH2O) or higher pressures (9-11). The primary outcome was extubation failure within 7 days, which was defined as the FiO2 increasing by 20%, or a resp acidosis (PCO2 >60 and pH <7.20), bad apneas or urgent need for re-intubation. This is their poster (from the PAS website), which shows there were many fewer extubation failures using higher CPAP, and no difference in other adverse outcomes.
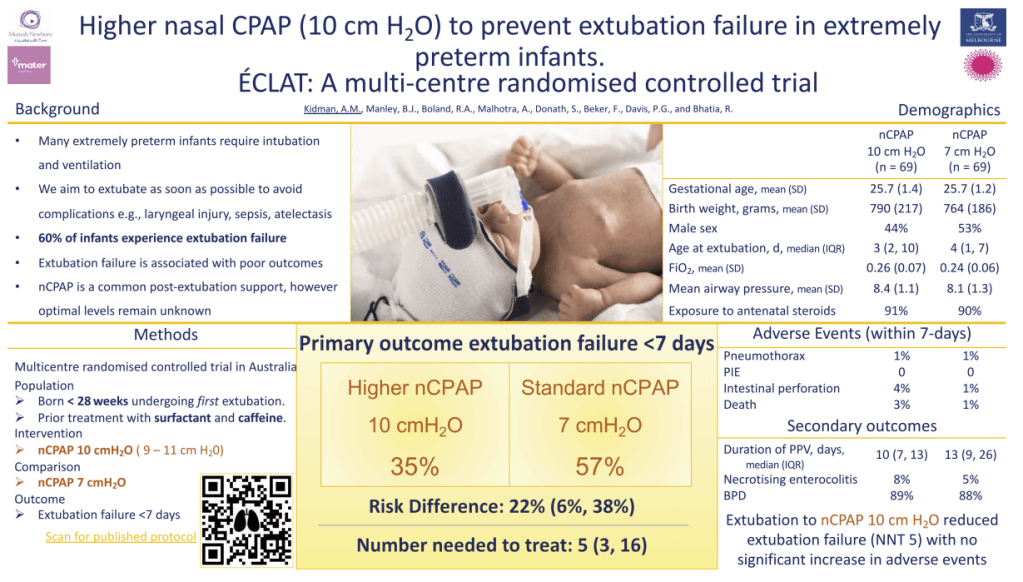
With 69 per group they were not powered for many other outcomes, and it is interesting that the incidence of BPD is very high in both groups, but I don’t know what definition this was using, I would guess it included mild BPD.
Would you like a side order of budesonide with that surfactant?
The results of the Taiwanese trial (sadly lacking an acronym) which randomized infants to surfactant alone (Curosurf) or poractant with budesonide (0.25 mg/kg/dose) and allowed up to 3 surfactant doses in each group, were presented. To be eligible babies were <1500 g at birth and required intubation in the delivery room or within 4 hours after birth. There were slightly over 300 babies in the trial, and the primary outcome was “BPD or death”. The average GA was about 27.5 weeks in each group. Mortality was similar in the 2 groups at close to 10%. 60% of the surviving controls developed BPD, and 38% of the surviving budesonide babies, with, also, more mild cases in the steroid group, and more severe cases in the controls.
It looks like the steroids were more effective in the larger more mature babies, secondary analysis by weight stratified subgroup doesn’t shown an effect in the <750 g babies, but there were less than 40 per group who were this small, and very little power. Blood pressures were higher in the treated group, suggesting systemic absorption, which you would expect. Alan Jobe has shown in preterm lambs that the majority of administered budesonide enters the systemic circulation, less than half being left in the lungs. I didn’t see any data on late-onset sepsis, and long term follow up will be important, as it is very early steroid use which has been most strongly associated with cerebral palsy in previous studies of systemic use.
None of the previous trials of steroids which have shown a decrease in BPD, and which have followed the babies after discharge, have shown improved long term respiratory health. Long term respiratory outcomes will be essential.
This looks encouraging, and I know some centres are doing this already, we will discuss in my centre, but I’m not sure I’m quite ready to expose every intubated VLBW to steroids on day one. Awaiting the PLUSS trial with bated breath.
Optimistic for long term pulmonary outcomes
Peter Dargaville presented the long term pulmonary and developmental outcomes of the OPTIMIST trial of minimally invasive surfactant treatment. In that trial 488 babies of 25 to 28 weeks gestation were randomized to MIST or ongoing CPAP or nasal IMV (PEEP of 5 to 8) if they needed 30% oxygen or more in the first 6 h of life. The primary outcome was “death or BPD”; there were slightly more deaths in the MIST group (10% vs 8%) but less BPD among survivors, 37% vs 45%. “Death or BPD” was therefore a bit less frequent with MIST (44% vs 50%) but was “not statistically significant”.
The follow up of that trial up to 2 years of age gave this CONSORT flow diagram, showing that up to 2 years the mortality was very similar. For various reasons, especially COVID, in person follow up was only the minority, but they had on-line questionnaires filled in by parents, and all the developmental outcomes were similar between groups. The proportion of babies with language or cognitive delay was identical between groups.
In contrast the respiratory outcomes all look better in the MIST group.
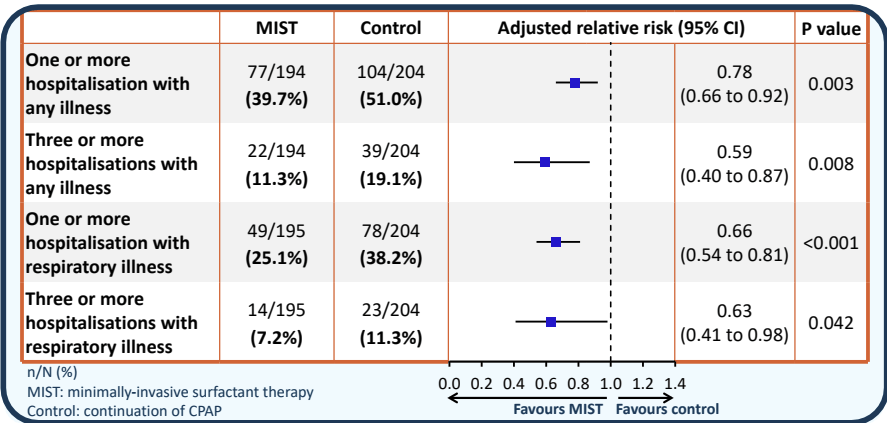
There were questions about whether this was really a trial of early versus late surfactant, but as Peter rightly said, it was not! Control group babies did not all get surfactant, babies in both groups were intubated for surfactant if they reached 45% (MIST was not allowed) and after intubation further therapy was according to usual practice, so INSURE, or more slow weaning, or whatever your usual was were OK. Most babies were expected to be on caffeine, but it was not mandated by the protocol.
This is one of very few studies of respiratory interventions which seems to show clear long term respiratory benefits, beyond just the presence of a BPD diagnosis. 72% of the controls got intubated within 72 hours of birth, compared to 37% of the MIST babies.
MIST in the delivery room, with coffee!
Anup Katheria presented another trial of thin catheter administration of surfactant, this time being randomized in the first hour of life, once babies of 24 to 29 weeks were stabilised on CPAP, and had received caffeine. Controls also received caffeine, but no surfactant, 180 babies were randomized and the main outcome was need for intubation, determined by needing over 40% O2, or a respiratory acidosis or bad apneas. Babies with less-invasive surfactant and caffeine required intubation in the first 72 hours of life 23% of the time, compared to 53% of the time for the caffeine only group. The surfactant group did get their caffeine a bit earlier than the controls (at 52 minutes of age compared to 70 minutes). Death before discharge was rare in this study (only 3 babies died, all in the control group), and there seems to be less BPD in the surfactant group, 26% vs 39%, although the study was not designed to have power for the BPD diagnosis, it looks like there probably was an effect.
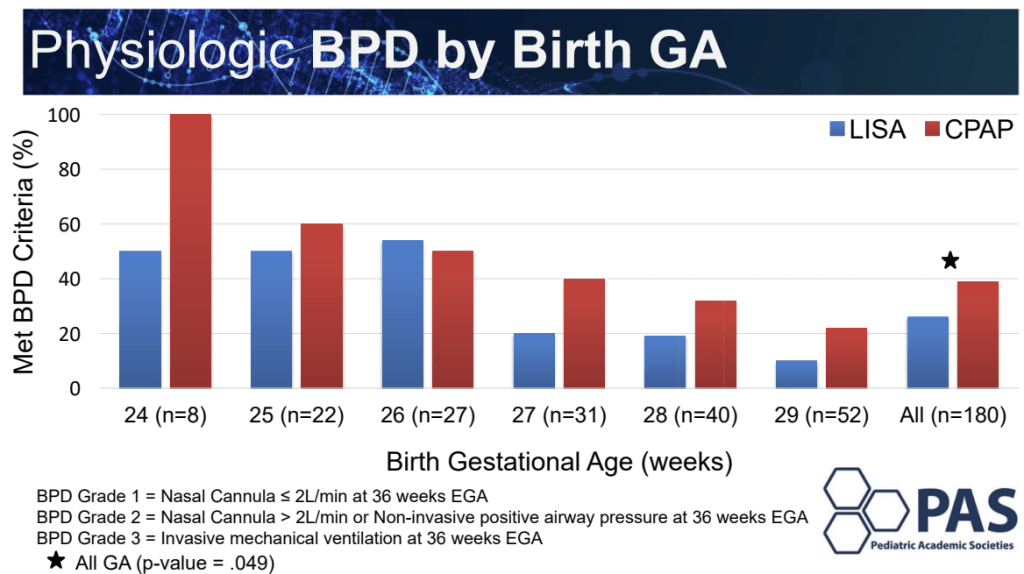
Anup Katheria helpfully listed the differences between his study and OPTIMIST

I really hate the idea of performing laryngoscopy without analgesia, and sedative premedications were not allowed in the OPTIMIST trial. I don’t know if they were allowed in CaLI, but if we can avoid intubation completely in a large number of LISA/MIST babies, which these survival curves below demonstrate dramatically, and therefore avoid prolonged desaturations, bradycardias and multiple intubation attempts, then maybe an un-premedicated less-invasive surfactant in early life will reduce, overall, the amount of pain experienced.

Putting things together
It is starting to look like very early minimally-invasive surfactant administration, perhaps routinely in the DR, and with very early caffeine administration, is the way to go. Not because it decreases “BPD” but because it seems that long term respiratory health is improved by avoiding intubation in the first few days of life (long term outcomes of SUPPORT and now OPTIMIST)
Perhaps early less-invasive surfactant should be co-administered with budesonide, but we should carefully consider that the NEUROSIS trial, of repeated budesonide inhalations starting during the first 24 hours, showed a small excess of mortality in the budesonide group. OPTIMIST also shows a small excess of mortality with budesonide. In addition, budesonide when mixed with surfactant enters the systemic circulation, and very early systemic steroids in preterm infants increase motor delay and cerebral palsy.
Of all the previous steroid trials that have shown reduced BPD and have also reported long term respiratory outcomes, I don’t think there is a single one which has shown improved longer term respiratory health.
Of course those studies are all contaminated by frequent treatment of control babies with steroids, so long term outcomes may be no different for that reason. On the other hand, potent systemic Steroids may decrease inflammation and thus lead to fewer babies needing oxygen at 36 weeks, but they also impair lung growth and interfere with lung development. I would like to see some evidence that long term respiratory health is improved before treating all babies receiving surfactant with budesonide, not to mention some evidence that there are no adverse effects on neurologic or developmental outcomes.








Hi Keith:
I too am beginning to believe that early caffeine along with our usual surfactant protocols will demonstrate some long term benefit. I am (as you are) concerned about the long term effects of steroids in growth and development. We might be successful with the need for less ventilatory support, but may be burdened with more chronic problems later on. Best to Annie and the twins.
Be safe…..but adventurous.
G
Pingback: Early routine surfactant, method and outcomes | Neonatal Research