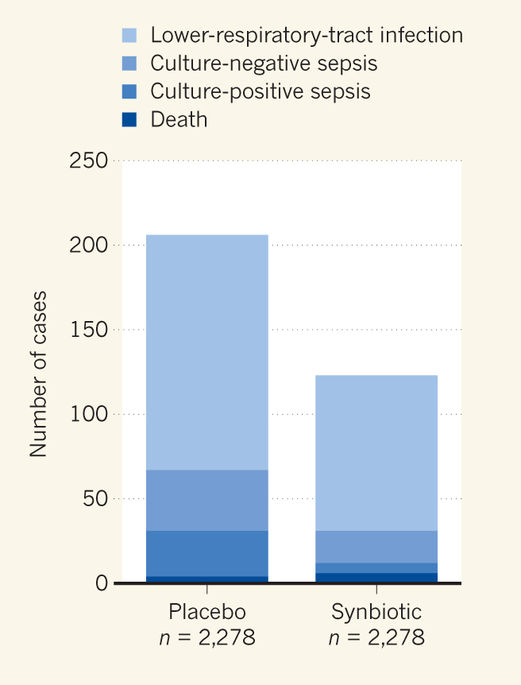
This post is a bit of a catch up, to mention articles that are worth reading, but which I didn’t have time to write a full post about, in particular studies that could have a positive impact on clinical care.
Let’s stop monitoring gastric residual volumes… please!
Measuring how much liquid is left in the infant’s stomach, just before the next feed, is a practice based on good intentions, but no evidence. Good-intention-based-medicine has, unfortunately, often been proved ineffective, or even harmful, especially when compared to evidence based medicine. This study from Israel (Riskin A, et al. The Impact of Routine Evaluation of Gastric Residual Volumes on the Time to Achieve Full Enteral Feeding in Preterm Infants. The Journal of pediatrics. 2017;189:128-34). showed the same as other studies, that the only clinically detectable impact of measuring gastric residuals is to slow down how fast babies are progressed to full feeds, without any clinical benefit.
If you give babies more nutrition, they grow more.
One of many studies that show that being more “aggressive” in early nutrition has positive impacts on weight growth, but also head circumference growth, and, perhaps, long term body composition.
Genoni G, et al. Nonrandomised interventional study showed that early aggressive nutrition was effective in reducing postnatal growth restriction in preterm infants. Acta Paediatr. 2017;106(10):1589-95. Approaching recommended standards for nutritional intakes improves usual measures of postnatal growth restriction, which are based mostly on weight.
The word “aggressive” bugs me here, the idea that trying hard to approach an intra-uterine delivery of nutrients, and approximate intra-uterine growth is somehow “aggressive” strikes me as wrong-headed. If we are to reduce the obvious negative impacts of being born far too soon, we must ensure that corporal and cerebral growth approaches normal values; not just in terms of quantity, but also quality. You can certainly have quantity without quality, but can you have adequate good growth, without enough overall weight gain?
If they grow more, are they smarter?
The next study addresses some of the same issues: Raghuram K, et al. Head Growth Trajectory and Neurodevelopmental Outcomes in Preterm Neonates. Pediatrics. 2017. Infants who had better head growth in the neonatal and the post-discharge follow up period had better developmental outcomes, Infants with poorer head growth in the neonatal period, up to discharge, were affected (i.e. had poorer developmental outcomes), and their head growth correlated with their body weight increase.
I think the first barrier to overcome is to ensure that weight, head, and length growth are close to normal intra-uterine values, then we need to evaluate what that means for details of body composition and developmental outcomes. I will come back to this issue soon!
At last, something better than amphotericin B?
This planned phase 3 study was stopped early because of inadequate numbers. Which is a great shame as we really needed to know if there was something better than AmphoB, which there probably is. In this study, micafungin was as effective as Amhpotericin B, and did not have more adverse events. Benjamin DK, Jr., et al. A Phase 3 Study of Micafungin Versus Amphotericin B Deoxycholate in Infants with Invasive Candidiasis. Pediatr Infect Dis J. 2018.
I have often seen minor adverse effects of Ampho B, and doses are often limited because of the concern about adverse events, most AE’s are minor, but a safer, equally effective antifungal would be a great addition. The echinocandin group, including micafungin, seem to be good candidates, with known pharmacokinetics, and a good safety profile. This phase 3 comparative trial showed little difference in efficacy or safety, but was underpowered for both, with only 30 babies in total (2:1 micafungin:amphotericin). Hats off to Danny Benjamin for trying to get this done, but a great problem in our community that we cannot do adequately powered trials to answer important question like this.