The dose of caffeine that we used for the CAP trial was the dose that was being widely used at the time. It seemed to be a safe dose, that did not require serum concentration surveillance, but was not based on a large number of high quality dose ranging trials, indeed it was identical to the doses used in the very first publication of caffeine use in the preterm infant, published in 1977 by Jack Aranda, from Montreal (the McGill side of the mountain). (Aranda JV, et al. Efficacy of caffeine in treatment of apnea in the low-birth-weight infant. J Pediatr 1977;90(3):467-72), they used 20 mg/kg of caffeine citrate as a load, and 5 to 10 mg/kg/dose once or twice a day, starting 48 to 72 hours after the load.
The maintenance dose gradually stabilised over the years, although there have been repeated questions about whether it is the optimal dose. There were some pharmacokinetic studies, but few quality pharmacodynamic studies, those that existed rarely used objective quantification of apnoea frequency, which is essential to be able to say much about the impacts on apnoea, nursing records of apnoea spells being notoriously unreliable.
The CAP trial remains the standard, which showed an improvement in medium term, and very long term, outcomes after caffeine use among infants of less than 1250 g birth weight, less than 10 days of age started on caffeine (or placebo) because the attending physician thought they needed caffeine. The dose in that trial was 20 mg/kg of caffeine citrate as a load, and 5 mg/kg daily, that could be increased to 10 mg/kg of caffeine citrate daily if the physicians thought the baby needed more. I submitted an abstract to a PAS meeting which showed that babies who had the dose increase had the same advantage of caffeine as those who remained on the initial dose, which I never fully published (sorry!) but I think was reliable information that the higher dose of caffeine was safe.
Clearly if some caffeine is good, then it is possible that much more caffeine might be even better. If we can keep babies extubated for longer periods of time, and knowing that several animal models show brain protective effects of caffeine, then what dose of caffeine should we give?
In this study in 3 day old rats, for example, the animals received 20 mg/kg/d of citrate for 5 days starting the day before a classical carotid-ligation-hypoxia model, and they had less white matter injury. In another study in newborn mice with an IVH model, caffeine at 20 mg/kg/day for 3 days started after the IVH reduced brain injury and brain atrophy. In another study Jack Aranda returns to caffeine 45 years after the first publication (!), comparing neuroprotection in newborn rats who received a dose similar to the usual human preterm dose (20 mg/kg load of Caffeine citrate followed by 5 mg/kg/d) or a larger dose of 80 mg/kg load followed by 20 mg/kg/d for 12 days. They showed similar neuroprotection with the 2 doses.
But before we ramp up the caffeine dose too far, remember the results of the pilot trial from St Louis, which randomized babies to get 80 mg/kg as the loading dose, starting in the first 24 hours of life, compared to 20 mg/kg in the standard dose group. The 80 mg/kg was actually given over 36 hours, as 4 doses, 40 mg/kg as the initial load, 20 mg/kg 12 hours later, then 10 mg/kg 24 and 36 hours after the first dose, the controls received 20 mg/kg then 10 mg/kg 24 hours later. Both groups in that small pilot (n=37 per group) received the same maintenance dose 10 mg/kg/day of caffeine citrate. The results of that trial showed a greater seizure burden with high dose compared to standard dose, and the high dose babies also had more cerebellar injury; fortunately the 2 year and 5 year outcomes were very similar between groups.
This was actually a slower load than in the previous trial by Steer et al who gave a 80 mg/kg bolus over 15 minutes, and then a maintenance of 20 mg/kg/d compared to their controls who got 20 mg/kg load then 5 mg/kg/d. In that study, the 240 babies of <30 weeks gestation were a little older (2 to 12 days of age, average 4 days) when enrolled and the study was designed to look at extubation failure. The primary outcome, extubation failure was less frequent in the high dose group, and there was a little less BPD in the high dose group 34 vs 48% RR 0.72 (95% CI 0.52-1.01). The medium term outcomes at 1 year of age tended to be better in the high dose group, and are described in more detail in this publication. The developmental quotient from the Griffiths scale is a slightly higher in the high dose group, but I don’t see any publication with later follow up.
There are a couple of smaller studies randomizing babies to higher doses of caffeine. In one of them, published in Chinese so I can only read the English abstract, 162 ventilated infants <32 weeks were randomized to different maintenance doses, they all received 20 mg/kg load before 6 hours of age (presumably of caffeine citrate) then either 5 or 10 mg/kg (presumably the daily dose of caffeine citrate); it appears that the higher dose group were less likely to fail extubation, which was mostly because of apnoea. An Egyptian trial (fortunately for me published in English) randomized 120 ventilated babies <32 weeks to receive either a load of 40 mg/kg of caffeine citrate and a maintenance of 20 mg/kg/d, or a load of 20 and maintenance of 10. They showed less extubation failure in the high dose group, but there is no longer term follow up. Another study from China randomized ventilated infants <30 weeks gestation who were over 48 hours of age and thought to be within 24 hours of an extubation attempt to a maintenance dose of either 5 or 10 mg/kg of caffeine citrate starting 24 hours after the load of 20 mg/kg, which was the same in each group. Extubation failure was decreased with the higher maintenance dose.
Putting this together to me this suggests that very early, very high loading doses of caffeine might be risky, that increasing maintenance doses to 10 mg/kg/d is probably safe and beneficial, with an increase in successful extubation and less apnoea, but limited long term outcome data. Increasing the loading doses after the first couple of days of life probably improves extubation success, but without more safety data I would be reluctant to use very high loading doses as a routine.
One of the particular features of caffeine kinetics is a dramatic increase in clearance as babies approach term. Caffeine is mostly filtered by the kidneys unchanged in early postnatal life of the preterm, and there is a gradual development of various pathways of demethylation with maturation, acetylation probably develops even later. The half life, as a result, is often over 100 hours in the early preterm period, falling to about 4 to 6 hours in an adult.
If we are planning to give caffeine to babies who are more mature, as they approach term, doses will therefore probably have to be adjusted. A trial which randomized 95 preterm babies who were stopping caffeine at >33 weeks, to either usual care without caffeine or to restart it 5 days later with a loading dose of 20 and maintenance of 6 mg/kg showed that the babies had fewer intermittent hypoxic spells in the caffeine group, until they reached 37 weeks PMA, by which time there was no longer much impact of the caffeine, I think this may have been partly because of a lack of power, as the controls were having fewer spells, but may also be because caffeine clearance was rapidly increasing and the infants needed more. The authors of this study therefore enrolled a second cohort of 27 similar babies who received higher doses, which they compared to the controls from their first study. The doses in the 2nd publication were started just 24 hours after stopping clinically required caffeine, and were 10 mg/kg/d of caffeine citrate, increased at 36 weeks PMA to either 14 or 20 mg/kg/d, decided by random allocation. The doses were chosen to try and maintain salivary caffeine concentrations at above 20 microg/mL, thought to be a reasonable therapeutic target for efficacy and safety. In this trial there was a reduction in intermittent hypoxic spells compared to the controls from the previous study.
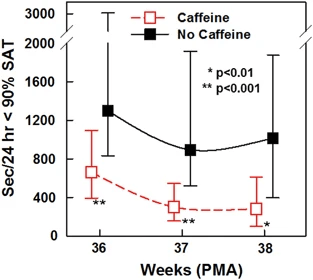
In a small short term study like this it is not possible to say whether there was a clinical benefit to the babies of having fewer intermittent hypoxic spells. As far as I can see there is no long term follow up of these babies published, but it would have very little power anyway.
All of which is a preamble to a newly published study from Auckland examining the use of caffeine in late preterm infants. We don’t normally worry about these babies in terms of apnoea of prematurity, even though they clearly very commonly have apnoeic spells, most of the spells are brief and resolve spontaneously. However they are accompanied by episodic hypoxia, and repeated hypoxia and re-oxygenation leads to an oxidative stress, which might have adverse long term effects. We also know that the neurodevelopmental long term outcomes of late preterm babies are different to those at term, with more cerebral palsy, and more schooling difficulties.
The new study was to determine a dose that decreases intermittent hypoxia in late preterm infants (Oliphant EA, et al. Caffeine to prevent intermittent hypoxaemia in late preterm infants: randomised controlled dosage trial. Archives of Disease in Childhood – Fetal and Neonatal Edition. 2022:fetalneonatal-2022-324010); babies born at 34 to 36 weeks were randomized to placebo or one of 4 doses ranging from 5 to 20 mg/kg/d, around 25 babies per group, with the first dose in each case being the double of the maintenance. Caffeine (or placebo) was continued until term, including after discharge, and the primary outcome was the number of intermittent hypoxic spells on an oximetry recording performed at 2 weeks after enrolment.
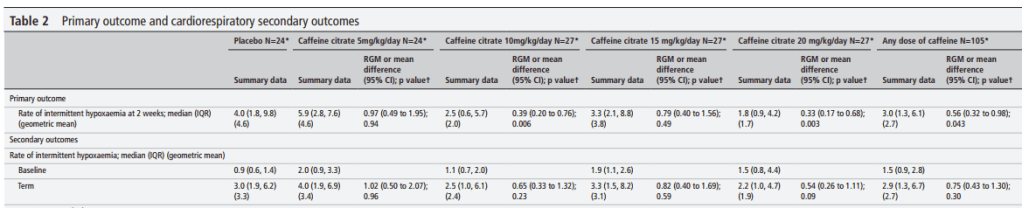
They showed fewer events at 2 weeks of age with the caffeine dose of 10 or 20 mg/kg/d compared to placebo (but not with 15 mg/kg/d), heart rates and episodes of tachycardia were increased in the caffeine groups. Presumably the lack of effect of the 15 mg/kg dose is just a random effect, as the groups were relatively small and the frequency of intermittent hypoxia spells is very variable.
This study confirms that to have an impact on respiratory drive and hypoxic spells near to term, the doses required are substantially higher. What it does not tell us is whether we should be doing this!
If you are caring for an individual child with troublesome clinically important apnoeas who is approaching term, and you want to treat with caffeine, you will need a higher dose to reduce the number of spells, because of the changes in metabolism.
Routine treatment of babies as they approach term is another question entirely. A higher dose than previously used will be necessary IF prevention of intermittent hypoxia spells near to term can be proven to be important. We have known for years that very preterm babies often continue to have multiple apnoeic spells as they approach term and hospital discharge (Barrington KJ, et al. Predischarge respiratory recordings in very low birth weight newborn infants. J Pediatr. 1996;129(6):934-40). We also know that there is a statistical correlation between more days with apnoeic spells and worse neurodevelopmental outcomes. Janvier A, et al. Apnea is associated with neurodevelopmental impairment in very low birth weight infants. J Perinatol. 2004;24(12):763-8.
What we need to know is whether it is safe and effective to give high doses of caffeine to very preterm infants as they approach term to reduce such spells. Even if, as seems likely, we can reduce intermittent hypoxia with higher doses of caffeine with routine continuation towards term, does it actually improve longer term outcomes. Even more important in some ways, given the very much larger numbers of at-risk infants, is it safe and effective to routinely treat late preterm infants to reduce their frequent hypoxic spells?
I am sure Dr Alsweiler and colleagues in Auckland are hoping to follow up the Latte Dosage trial with a large multicentre RCT examining long term impacts of high dose caffeine in late preterm infants, lets hope they get the funding. We tried a couple of times to get NIH funding for a trial in very preterm babies as they approached term, but the trial, as it was designed, was hugely expensive and never got high enough priority. A simpler pragmatic trial would be very valuable, and could inform our community about what to do with caffeine for the preterm infant approaching term.








