A new study from Finland this time, raises similar concerns to those from a Canadian study. (Raikkonen K, et al. Associations Between Maternal Antenatal Corticosteroid Treatment and Psychological Developmental and Neurosensory Disorders in Children. JAMA Netw Open. 2022;5(8):e2228518). It is an analysis of the same dataset that the authors previously published about; in that previous study they lumped together developmental and behavioural disorders, and did not analyse neurosensory difficulties. Using the same enormous linked databases with over half a million children, of whom just over 2% had been exposed to antenatal steroids (presumably all fluorinated steroids for lung maturation). The authors state that they have previously validated that these cases are all indeed betamethasone in threatened preterm delivery. This is important of course, because people take steroids for all sorts of reasons, even during pregnancy, and most are metabolized by the placenta and have no direct fetal or neonatal impact of note.
This time they also differentiated between those who delivered at term and those who were actually preterm. The graphic below shows that most of the outcomes were similar between preterm exposed and non-exposed to steroids, even though the actual gestational age at birth was lower in the preterm babies who had been exposed to steroids (mean 32.8 (SD3.0) vs 35.5 (SD1.7) weeks). Among the babies who delivered at term the exposed and unexposed had very similar GA (39.3 vs 40.1) and other characteristics, but had more adverse outcomes, in just about every domain, with cerebral palsy being the most striking.
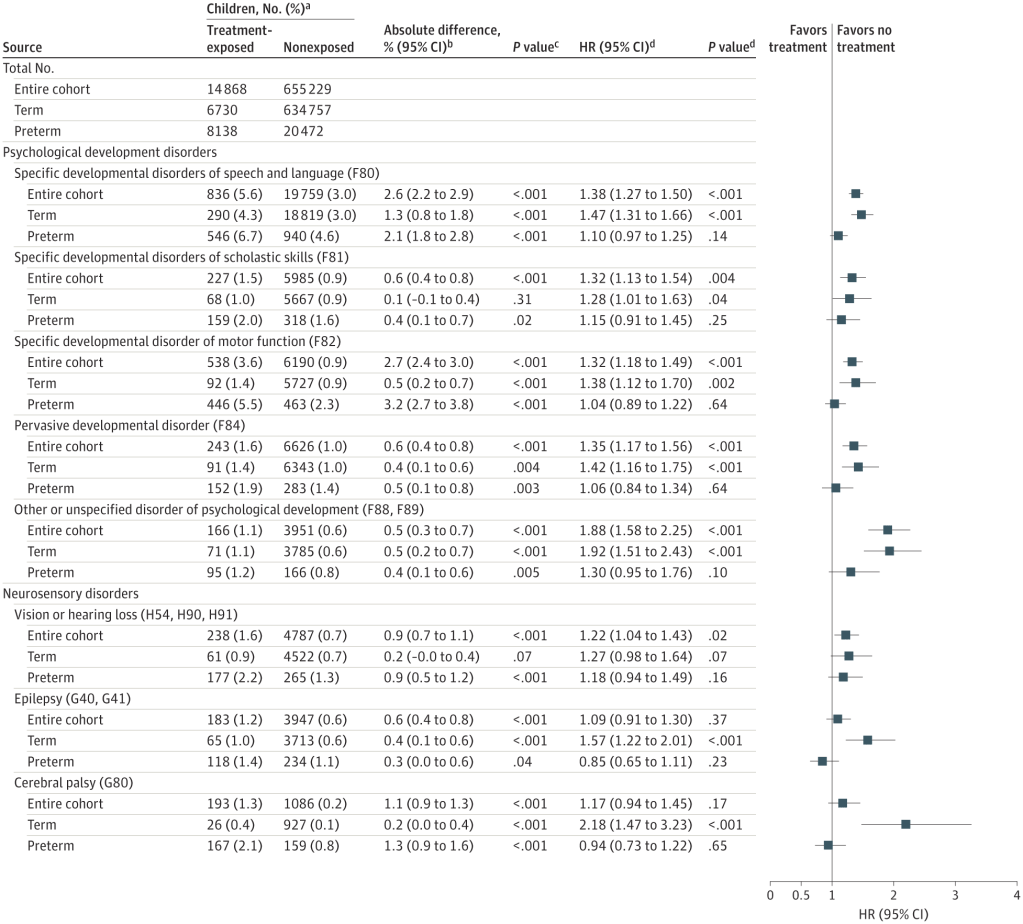
The absolute risks are small, however, for CP, for example, the Hazard Ratio is over 2, but the absolute percentages are 0.4 compared to 0.1.
These results are not dissimilar to data from Ontario (Aviram A, et al. Antenatal corticosteroids and neurodevelopmental outcomes in late preterm births. Arch Dis Child Fetal Neonatal Ed. 2022;107(3):250-5) another database study showing that having received antenatal steroids is associated with an increase in billing codes that reflect suspected neurocognitive disorders.
What should we do about these data? I think that we should be more circumspect about steroid use in the late preterm, especially after 35 weeks when the benefits are small. Also we need to find ways to better target steroids to those who are more likely to deliver preterm.
Maybe we should also be reducing the dose? Well…. maybe not (Schmitz T, et al. Neonatal outcomes for women at risk of preterm delivery given half dose versus full dose of antenatal betamethasone: a randomised, multicentre, double-blind, placebo-controlled, non-inferiority trial. The Lancet. 2022;400(10352):592-604) In this multicentre French RCT, over 3000 mothers with threatened preterm delivery who had received their first dose of betamethasone and were less than 32 weeks gestation were randomized to receive either placebo or betamethasone for the second dose. The primary outcome of the study was the need for surfactant treatment, I guess that is reasonable as a primary, it is probably not the outcome I would have chosen, but I am not sure what would have been!
About 60% actually delivered preterm in the study, only 30% prior to 32 weeks gestation, and another 10% prior to 34 weeks. About 20% in each group required surfactant, and the 95% confidence intervals for the difference in surfactant requirements crossed the non-inferiority boundary, leading the authors to conclude that they were unable to show non-inferiority.

Although this is an important, well-designed study, I do have doubts as I said, about the primary outcome. We don’t really give steroids to avoid giving surfactant, but for all the other benefits on maturation, and most importantly a reduction in mortality in the most immature infants, but a study designed with a mortality outcome would have to be absolutely enormous, so overall, I think needing surfactant is a reasonable proxy outcome.
What about the other important neonatal outcomes?
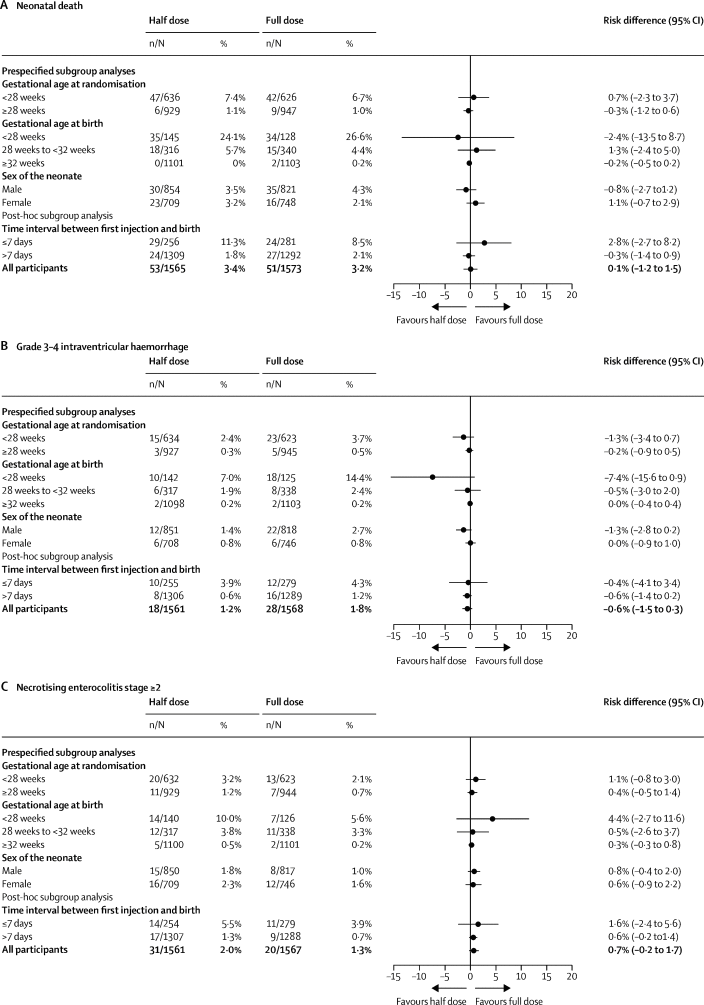

As you can see there is no clear difference in any of the outcomes, with the exception of the combination outcome among infants who were born within a week of getting the steroids.
It looks like the lower dose is just about as good as the full dose, but, being stringent in our interpretation, they have not shown non-inferiority, the outcome was a little bit more frequent in the low dose dose group, and is consistent with a poorer efficacy. Of note there were only about 250 in each group that delivered within 7 days of receiving steroids.
I was encouraged to read that the study was funded for long term outcomes, with a 5 year assessment including WPPSI and NEPSY subsets and a neuro exam. Also that a Canadian and Australian trial is underway SNACStrial.com with short and long term outcomes being investigated.
It is going to take a while, but we will eventually have good quality data about whether halving the dose of betamethasone is as effective, and potentially safer, than the current dose. If you remember, the currently used dose was derived directly from the original Liggins studies in sheep, and there has never been, until these new trials, any dose response data. It is about time.








