Many, many years ago, when I was a young trainee physician, we learned almost everything “on the job”. I can’t remember the first patient I intubated, but there were no mannequins, and no simulations, the phrase “see one, do one, teach one” was perhaps an exaggeration, but not far from the truth. When I read, recently, the hilarious book ‘This is Going to Hurt’, by Adam Kay which recounts his time as s junior obstetric trainee doctor in England, it seems that a similar approach continued when he was training, fairly recently.
I don’t remember the first patient I intubated, it was certainly an adult, as I did jobs as a House Officer (intern), then a year as a Senior House Officer in adult medicine before switching to paediatrics. During that year I would often institute intensive care for the most critically ill patients, I remember inserting peritoneal catheters to start dialysis, putting in central lines to start intracardiac pacing, and intubating several patients. The first one, or maybe two, intubations were probably done with an anaesthetist standing by my shoulder coaching me. By the time I did my first neonatal job I was asked if I was able to intubate, and, as I answered in the affirmative, I was given first option on the intubations that occurred in the NICU during my calls. In those days we also intubated in the delivery room all babies born with thick meconium in the amniotic fluid, as we were convinced this would reduce the risks of severe meconium aspiration syndrome. We would repeatedly intubate and suck on the tube as we removed it, until the fluid that returned was clear, so sometimes needing 3 or 4 intubations and sometimes even more. We even tried to stop the babies breathing vigorously as we did the intubations! This meant that I rapidly became an expert at endotracheal intubation in larger babies, and gradually in smaller infants also.
Returning to the present, we now know a lot more about physiologic responses to intubation, and the adverse impacts of failed intubations and multiple attempts (Singh N, et al. Impact of multiple intubation attempts on adverse tracheal intubation associated events in neonates: a report from the NEAR4NEOS. J Perinatol. 2022). For example, having multiple attempts at endotracheal intubation in the delivery room increases the risk of intraventricular haemorrhage (Sauer CW, et al. Intubation Attempts Increase the Risk for Severe Intraventricular Hemorrhage in Preterm Infants-A Retrospective Cohort Study. J Pediatr. 2016;177:108-13), and many babies desaturate severely during intubation. It is critically important to make intubation as safe as possible for at-risk babies. At the same time we have to train new young paediatricians, many of whom will eventually work in hospitals with delivery rooms, where they will be responsible for neonatal resuscitation, and may provide coverage for level 2 neonatal units where intubation skills may be important. We have therefore, a dual responsibility, both to future babies, to ensure that our trainees can care for them adequately, but, most importantly, to the fragile babies currently in our NICU.
My colleagues and friends, the 3 authors of this study, Michael, Christian and Ahmed, (Assaad MA, Lachance C, Moussa A. Learning Neonatal Intubation Using the Videolaryngoscope: A Randomized Trial on Mannequins. Simul Healthc. 2016;11(3):190-3) have been performing a series of studies of the best way to teach intubation skills. Residents now intubate mannequins before they ever touch a baby, and they have a refresher course with a mannequin and the videolaryngoscope at the start of their first NICU rotation. They are supervised performing their first real-life intubations with video laryngoscopy, (Moussa A, et al. Videolaryngoscope for Teaching Neonatal Endotracheal Intubation: A Randomized Controlled Trial. Pediatrics. 2016;137(3):1-8), and most subsequent intubations in the NICU are performed by trainees using the videolaryngoscope. In addition, all intubations in the NICU (except in a major emergency) are premedicated, with atropine, fentanyl and succinylcholine (Barrington K. Premedication for endotracheal intubation in the newborn infant. Paediatr Child Health. 2011;16(3):159-71). Atropine reduces bradycardia, fentanyl reduces pain (and intubation is a very painful procedure) and improves physiologic stability, and succinylcholine shortens the overall duration of intubation (Barrington KJ, et al. Succinylcholine and atropine for premedication of the newborn infant before nasotracheal intubation: a randomized, controlled trial. Crit Care Med. 1989;17(12):1293-6) and reduces the number of attempts required.
Despite these standards, endotracheal intubation remains a procedure with high risk of desaturation, occasional bradycardia, and sometimes requires multiple attempts. Our group decided a few years ago that it wasn’t a good idea to have trainees performing their first intubations with the most fragile babies. We therefore restricted endotracheal intubation of the highest risk babies to only those who had demonstrated competence in the procedure with larger babies. Babies under 29 weeks gestation are only intubated by the “tiny baby” team members, which includes neonatologists and fellows, nurse practitioners, respiratory therapists who are members of the transport team, and residents. In order to be on the team, an intubator had to successfully complete at least 5 intubations on larger infants, 4 of which had to be with either 1 or 2 attempts. These criteria were entirely arbitrary. It was difficult to decide what the criteria should be as there was little previous information to base them on, and if we were too restrictive there would not be enough people around to ensure that there was an intubator on every shift! We have just published our experience with this approach, comparing pre- and post-institution of the tiny baby team. (Gariépy-Assal L, et al. A tiny baby intubation team improves endotracheal intubation success rate but decreases residents’ training opportunities. J Perinatol. 2022). We compared 3 periods, just prior to starting the team, a second period starting 6 months later, and a 3rd period starting 4 years later.
Here is an edited version of table 2 from the publication, showing the overall numbers and results of “ETI” endotracheal intubation, in the 1st 3 columns, then the results for the tiny baby team and the remaining infants. One thing you can see is the reduction in overall intubations, with more babies being managed non-invasively, a change which is most marked <29 weeks.
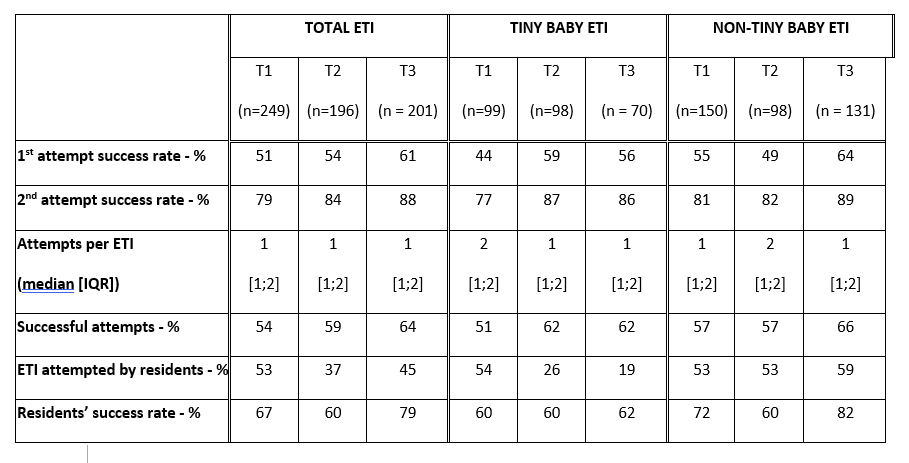
You can also see that the success rate, on first attempt, among the tiny babies increased when the team started and has remained higher, and the proportion needing more than 2 attempts dropped from 23% to just over 10%. Over these periods, if the first intubator was a junior trainee, they were only usually allowed one attempt, which is why the residents’ success rate is lower than the second attempt success rate in all categories. The number of intubations and the proportion performed by residents have both fallen overall, which has made it even more important to ensure that all forms of training are optimized for future paediatricians to be able to adequately perform the task. One change between T2 and T3 was that residents were prioritized for the intubations of babies >28 weeks. Coupled with enhanced simulation training, their success rate for the larger babies was improved in the most recent period.
This study wasn’t designed to look at the physiologic changes during intubation, but the NEAR for Neos registry has recently published data confirming that serious adverse events are more common with multiple attempts. (Singh N, et al. Impact of multiple intubation attempts on adverse tracheal intubation associated events in neonates: a report from the NEAR4NEOS. J Perinatol. 2022;42(9):1221-7). This is a multicentre registry with data contributed by many hospitals in the USA, Canada and Australia. Those data confirm that more than 1 attempt, and especially more than 2 attempts, are associated with serious adverse events. Severe “TIAE”, tracheal intubation associated events, include cardiac arrest and massage, airway injury, vomiting and aspiration, air leaks, and delayed recognition of oesophageal intubation.
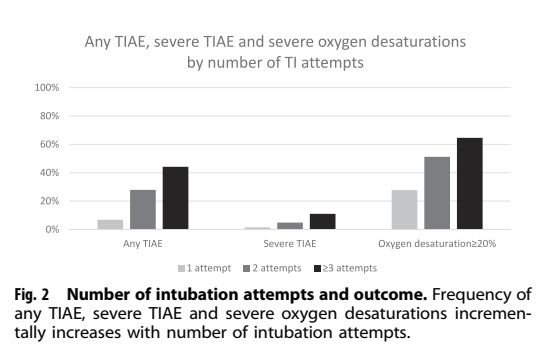
They confirmed that the requirement for multiple attempts was much greater among the most immature babies, and also when the intubator was a paediatric trainee, and was much lower when the baby had received a muscle relaxant. Overall 22% of the intubations required more than 2 attempts.
I was disappointed to see that of the over 6,600 intubations, 3,800 of which were in the NICU, only 2,760 received sedation and a muscle relaxant. Of those that did receive the optimal combination, only 16% needed more than 2 attempts. This is not the first time this group has reported the benefits of sedation with paralysis for reducing “difficult intubation”, and, of all the dozen or so reports I have found of the use of muscle relaxants during intubation, they universally show a reduction in adverse outcomes, depending on what they were measuring, either serious adverse events, desaturation, duration of attempts, or number of attempts. I don’t think there is a good excuse for not giving newborn infants requiring non-emergency intubation in the NICU an adequate pre-medication with a potent rapidly acting analgesic (either fentanyl or remifentanil are the best options) and a muscle relaxant (either succinlycholine or mivacurium, unless you want more prolonged paralysis in which case rocuronium) with atropine to prevent reflex vagally mediated bradycardia. In one recent publication it was reported that it took an median of 16 minutes for the babies to receive the premedications, I find that a little bemusing. We have a “crash cart” in the NICU with the medications easily available, pre-printed charts with the doses already calculated for each step of 100 g of weight, and all the equipment that may be required. Once I say I want to intubate a baby, the crash cart and additional nursing staff arrive, and the baby is often receiving the atropine within 3 minutes, there aren’t many intubations that are so urgent that they don’t receive our cocktail.
The other thing that you can do to improve stability during neonatal endotracheal intubation is to provide a flow of oxygen. My mentor, Neil Finer, was ahead of his time in many ways, he was among the first to study premedication for neonatal intubation, and one thing that was standard in his NICU was the oxyscope, a laryngoscope with an oxygen channel, to which an oxygen source was attached during intubation. I think “Oxyscope” was a trade name which may have been replaced by “Oxiport”, and I am not sure is still being manufactured, but it provided a fresh gas flow near the larynx which decreased desaturation. (Ledbetter JL, et al. Reducing the risks of laryngoscopy in anaesthetised infants. Anaesthesia. 1988;43(2):151-3). Although not using that commercially-produced blade, a much more recent publication (Steiner JW, et al. Use of deep laryngeal oxygen insufflation during laryngoscopy in children: a randomized clinical trial. Br J Anaesth. 2016;117(3):350-7) has confirmed that taping an oxygen cannula to a standard laryngoscope blade also works, and that a video-laryngoscope blade with an integrated oxygen channel exists, which also decreased desaturation in larger children during intubation. These laryngoscopes have only been studied with 100% oxygen flows, usually about 2 litres per minute, and there are of course concerns about brief episodes of hyperoxia that might be associated with their use. However, hypoxia and subsequent re-saturation is probably rather worse for the generation of free radicals than a couple of minutes of hyperoxia, and using a fresh oxygen flow into the pharynx of an apnoeic infant is unlikely to lead to much hyperoxia anyway.
Another way of providing apnoeic oxygenation during intubation is with the use of high-flow nasal cannulae (HFNC), I already posted about the SHINE study from Melbourne using high-flow, here is another trial, a pilot from Dublin (Foran J, et al. Nasal high-flow therapy to Optimise Stability during Intubation: the NOSI pilot trial. Arch Dis Child Fetal Neonatal Ed. 2022:fetalneonatal-2022-324649), as a pilot there were only 43 babies, and 50 intubations included. Infants (who were all premedicated, as in the SHINE trial, with atropine fentanyl and succinylcholine, also known as suxamethonium) had HFNC placed at 6 lpm with 100% oxygen, which is different to the SHINE trial who used a flow of 8 lpm at the same FiO2 as the infant was already getting and only increased to 100% if they desaturated. Another difference is that SHINE was just during the 1st intubation attempt, whereas in this trial they removed the cannulae after the first attempt and put them back if another attempt was required. This new trial had as the primary outcome the duration of desaturation below 75%, which was shorter in the preterm babies (median 29s vs 43s) and not much different in the term babies, as I said, this was a small pilot.
Here is the profile of the median saturations in the <34 week and >33 wk groups.
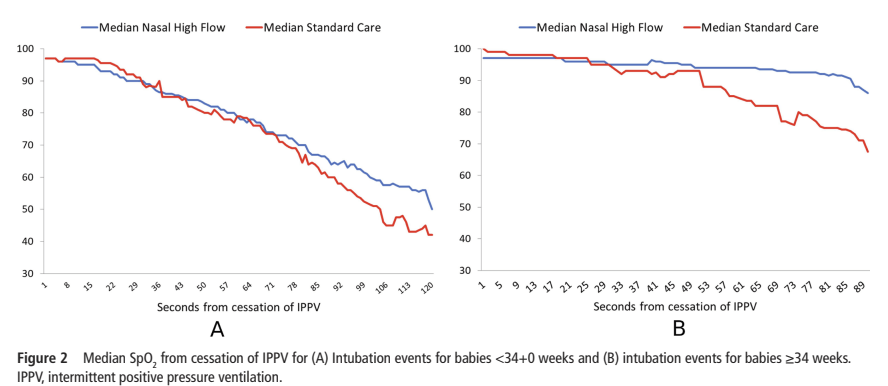
It seems to show two things, that the babies clearly desaturate faster without oxygen (!) and that someone should have stopped the intubation attempts before they got out to 120 seconds!!! The NRP used to state that an intubation attempt should take a maximum of 20 seconds, Neil Finer showed that was unrealistic Lane B, et al. Duration of intubation attempts during neonatal resuscitation. J Pediatr. 2004;145(1):67-70, that even experienced fellows took an average of 22 seconds, and Colm O’Donnell, when he was in Melbourne (O’Donnell CP, et al. Endotracheal intubation attempts during neonatal resuscitation: success rates, duration, and adverse effects. Pediatrics. 2006;117(1):e16-21), showed that the Australian trainees were slower, averaging 38 seconds for residents, and 32 for fellows. The 7th edition of the NRP handbook continues to state that “the steps of intubation should be completed within approximately 30 seconds”. That is clearly unrealistic, given that the average time for consultant neonatologists to perform a successful intubation in O’Donnell’s study was 25 seconds SD 17s. In experienced hands only about a half of intubations are completed in 30 seconds.
Most babies these days will have a pulse oximeter in place, and hopefully functioning, during an intubation, even in the DR. I think that after 30 seconds there should be an evaluation of the babies status, and if the baby is desaturating and the intubation is not completed by 40 seconds a decision whether to continue or interrupt the intubation, by someone other than the intubator, should be made. This is actually one way that I find the videolaryngoscope useful, I can see if the trainee has a view of the larynx, and is about to insert the tube, compared to the situation with the larynx briefly flying past the screen, and the tube tip heading for the dark hole of the oesophagus.
I don’t know if the combined oxyscope/video laryngoscope would be more or less effective than HFNC to reduce desaturation and adverse events during intubation, but I think someone should find out!
In summary, making endotracheal intubation safer for our most fragile patients requires the following:
- Adequate training of all intubators with simulation and video-laryngoscopy
- Step-wise introduction of intubators, with video-laryngoscopy, supervision, feedback and repeated training
- Ensure that someone with proven competence performs the procedure, by limiting intubation of the highest-risk patients to a restricted list of intubators.
- Universal premedication, including muscle relaxation, unless there is a contra-indication, and have procedures in place to administer with minimum delay.
- Apnoeic oxygenation, with HFNC, or perhaps an video-oxyscope
- Video-laryngoscopy, if you have access to a laryngoscope blade of appropriate size
- Ensure supervision of the baby, and their status, by someone who is empowered to stop the procedure if it is going wrong, or is taking too long.
- Feedback and further training whenever things go wrong, and even if they don’t.
No more “see one, do one, teach one”!
Even in our NICU, with most of this in place, over 10% of the very immature babies need more than 2 attempts to intubate, we have to find ways to do better than that, to reduce the number and the consequences of failed intubation attempts. During the study that I referred to, of the tiny baby team, we did not have a video-laryngoscope blade that worked for the extremely low birth weight baby, newer technology, and perhaps ever-more realistic high-fidelity mannequins, may help us to further reduce failure rates.









Thanks a lot Keith for all the important information.
What is your opinion and practice in premedication for minimal invasive surfactant treatment. Do you give the full cocktail?
That’s a really pertinent question, we certainly don’t use muscle relaxants! I hate the idea of laryngoscopy without analgesia, so at present we opted for ketamine, I don’t know if that is the best, we need more trials of that specific question.
Dear Keith we have developed premade intubation drugs and a dose banding that has now been used on over 1000 babies. They are kept on our unit in a locked fridge and we have done a pre and post human factors study to implement practice. The drugs can given with a double check in less than 5 minutes. Happy to share with you our guidelines. In terms of safety all 3 premedications have the same dose in mls.