When we consider using steroids in a preterm infant, ventilator dependent, with evolving lung disease, what outcomes are we most interested in? Survival, surely, is the first outcome that we want to improve, and secondly long-term pulmonary health. The adverse outcome we are most interested in avoiding is worsened development or adverse neurologic impacts.
Does it really matter if an individual baby still needs oxygen at 36 weeks post-menstrual age, that outcome is only of interest if it is a good predictor of respiratory health in the long term. Which it is not. When Andrew Shennan first proposed oxygen dependency at 36 weeks PMA as the definition of BPD, rather than the original definition of oxygen dependency at 28 days of age, it was because there seemed to be a good correlation with longer term respiratory symptoms after discharge. With the major advances in survival at extremely low gestational ages since then, the majority of our survivors of the earliest gestational ages still have some need for respiratory support, oxygen or positive pressure at 36 weeks, and many have respiratory problems ongoing after discharge.
More recent attempts to improve the usefulness of the diagnosis of BPD still concentrate on making the decision at 36 weeks, but why? Is it because many babies are often discharged shortly after 36 weeks, so, to make it a clean outcome for research, we like to have a yes/no outcome before the babies go home and start to be lost to follow up? That might make some sort of sense from a research design point of view, but we really should be studying things that have impacts on families and the future life of our patients.
The updated NICHD definition of BPD was shown in a publication from 2005 to be statistically significantly associated with readmission for respiratory illness, and with the use of respiratory medications up to 18 to 22 months corrected age. But statistically significant does not mean clinically useful; of babies in the NICHD database “without BPD” 24% were rehospitalised for respiratory indications and 27% received chronic respiratory medications. Among those with mild BPD the relevant figures were 27% and 30%, with moderate BPD, 33% and 41%, and those with severe BPD, 39% and 47%. There is therefore a significant trend to worse outcomes with worse grade of BPD, but the discriminatory power is not very good. One concern about these figures is that the use of chronic respiratory medications is very variable, as, in general, they don’t work! Diuretics, brochodilators and inhaled steroids are of questionable efficacy in infants with established BPD. Some physicians will use them more liberally than others, introducing variability that makes medication use problematic as an outcome variable.
A reduction in the frequency of BPD might, then, in a large study be an overall predictor of a probable improvement in respiratory health over the first year of life; a reduction in severe BPD being much more important than a reduction in mild BPD (even though many infants “without BPD” have respiratory problems in the first 18 months after discharge). So one could propose its use as a proxy for clinically important lung injury. But, importantly, an intervention which improves short term gas exchange and lung mechanics, but does not improve lung repair, could easily lead to a reduction in the proportion of babies with a research diagnosis of BPD, without any benefit on pulmonary outcomes in the longer term.
Systemic steroids reduce inflammation in the short term, improving lung mechanics and gas exchange, leading to fewer babies being diagnosed with BPD in many trials. But they also impair lung growth and development in animal models, and have never been shown to enhance repair in relevant models. It is possible, at least, that they could decrease the proportion of babies with “BPD”, but worsen longer term pulmonary health.
What are the impacts of postnatal steroids during evolving chronic lung disease of prematurity, on long term pulmonary health? The very depressing answer is that we have no clear idea. Despite more than 20 trials, including over a thousand very preterm infants, there is very limited data to determine whether giving a course of hydrocortisone or dexamethasone actually improves long term pulmonary health outcomes.
The Cochrane reviews of postnatal steroids in preterm infants don’t even have long term pulmonary health as an outcome. The longest term outcome they analyse is home oxygen, which is an important outcome for its impact on families, but misses a lot of respiratory morbidity.
Is there any evidence at all that postnatal systemic steroids improve long term respiratory health?
There is a small amount of information from some of the trials of dexamethasone (DEXA). The UK led dexamethasone trial published 3 year outcome data (Jones R, et al. Controlled trial of dexamethasone in neonatal chronic lung disease: a 3-year follow-up. Pediatrics. 1995;96(5 Pt 1):897-906), and found small differences in the outcomes they measured, but no clear advantage of DEXA. You also have to remember that over 40% of the placebo babies received DEXA at some point.
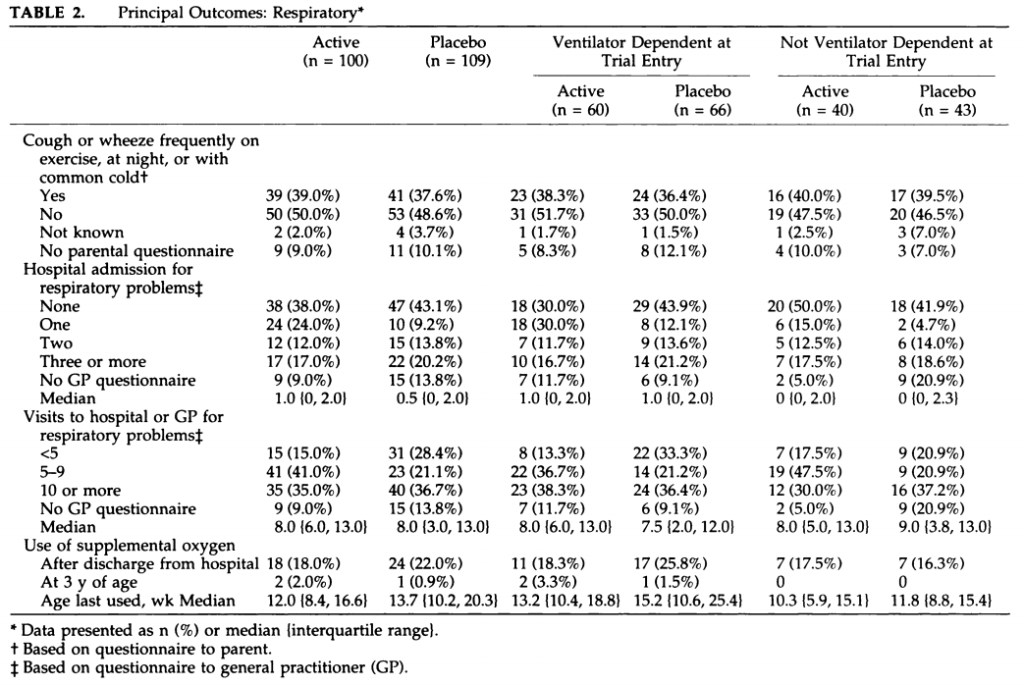
There are two publications of very tiny studies with some longer term respiratory follow up, which show either no difference, or an advantage of DEXA in terms of shorter home oxygen.
The Taiwan early dexamethasone trial was performed in the pre-surfactant era, dexamethasone was started within 12 hours and tapered off over 4 weeks in 262 ventilated preterm infants <2kg. The relevance to this discussion is that there was very little treatment of controls with DEXA (7 controls and 5 DEXA babies), and it was one of the first studies to confirm the neurotoxicity of DEXA, with lower BSID2 scores (both MDI and particularly the PDI) and much more cerebral palsy. Even though the results showed less oxygen requirement at 36 weeks, and thus less BPD (21% vs 36%), on 2 year follow up of 116 of the survivors, there were no differences at all in clinical respiratory outcomes : “There was no difference in the incidence of lower respiratory infections between the two groups (control group 13/59 vs dexamethasone-treated group 11/57). There was also no significant difference in rehospitalization for respiratory reasons during the first 2 y of life between the two groups (control group 18/59 vs dexamethasone-treated group 15/57)” and more extensive lung function testing also showed no differences of any note:
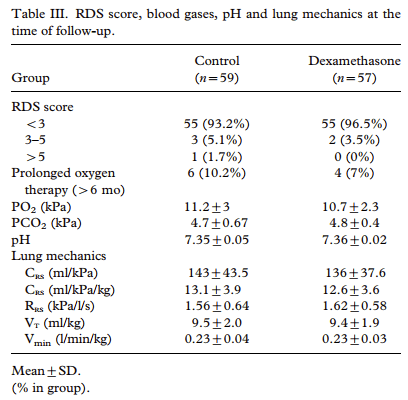
The DART trial enrolled infants much later in their hospital course, and did show acute impacts leading to reduced ventilatory dependency. However, almost all the babies satisfied BPD definition in both groups, at around 90%, and nearly half in each group went home on oxygen. Hospital readmissions and duration of home oxygen were very similar in the two groups.
One of the few trials in which controls were not allowed to receive open-label DEXA was the trial of O’Shea et al, in which a 42 day tapering course of DEXA was started at 15 to 25 days of age for VLBW infants who were ventilator dependent in more than 30% O2. 120 babies were randomized, and those who received DEXA were weaned faster from the ventilator, mortality was 7/57 with DEXA compared to 16/61, which was not “statistically significant” but is quite a big reduction in a small study, O2 dependency at 36 weeks was much lower in the DEXA group, 59% vs 82% in controls. The one year outcome of the study showed no difference in re-hospitalisation, which is the only longer term respiratory outcome reported (most re-hospitalisations are for respiratory reasons, but that was not specified in the publication). To recall, this was the trial that, more than any other because of the prohibition of DEXA use in controls, showed that DEX causes cerebral palsy. Definite CP in the controls was 7%, with DEXA that increased to 25%. Let us also remember that you don’t need a 42 day course of DEXA to increase the risk of CP, the 3 day course of 0.5 mg/kg/day studied by Shinwell et al, in 248 preterm infants with HMD started within 12 hours of life, increased CP from 14% in the controls to 46% with DEXA (25% of controls received open label steroids in this study). No data on respiratory long term outcomes was presented.
The very limited evidence that is available, therefore, shows no improvement in long term pulmonary health with DEXA, either early or late, despite a reduction in the diagnosis of BPD.
What about moderately early intervention with hydrocortisone? Use of this molecule between 1 and 4 weeks of age, has really only been studied in these two recent trials I have been discussing, STOP-BPD and the NICHD hydrocortisone study. Although there are clearly differences, there are enough similarities, I think to draw some conclusions. Together, they suggest that starting hydrocortisone will improve short term respiratory status, allowing extubation. There is a suggestion of a reduction in mortality, but as physicians have difficulty in avoiding systemic steroids in the sickest babies, this is not entirely clear. You would expect, however, that improving lung mechanics and gas exchange in the acute phase will prevent some respiratory deaths. Because of clinicians appropriate unwillingness to let babies die without a trial of steroids, it is impossible to prove from the current literature that hydrocortisone (or indeed DEXA) given moderately early do indeed decrease mortality.
The implications for practice, I think, are that steroids should be used to prevent mortality. For an infant who is at increased risk of respiratory death, use of steroids is justifiable, but which steroid and which dose are still uncertain. Initial treatment with hydrocortisone may be effective, and, if there is inadequate response, dexamethasone might be a more effective alternative but with known neurotoxicity issues.
There is currently little reliable evidence about the neurotoxicity of hydrocortisone in the developing preterm human brain. I can’t find a huge amount of animal data either, but there are certainly some studies which suggest that hydrocortisone may also have negative impacts on brain development, this one for example 😦Aden P, et al. Low-potency glucocorticoid hydrocortisone has similar neurotoxic effects as high-potency glucocorticoid dexamethasone on neurons in the immature chicken cerebellum. Brain Res. 2008;1236:39-48). It seems likely that DEXA has more negative effects on brain development that does hydrocortisone. Observational studies have shown more negative impact on MRI of DEXA compared to hydrocortisone, but with all the usual limitations of that type of evidence.
I think we should really question the use of steroids, DEXA or hydrocortisone, as a tool for getting babies extubated. Which is worse in the long term, a few more days of assisted ventilation, or a course of systemic steroids? In an intubated baby at low risk of dying, but high risk of developing BPD, hydrocortisone (or DEXA) will lead to earlier extubation, and may reduce the probability of having a BPD diagnosis, with no evidence that any important aspect of long term pulmonary health is improved.
Is there enough equipoise to perform another study? (And the willpower!) If we eliminate babies for whom steroids are already probably indicated (say a predicted mortality over 25% using the NICHD calculator, which remember was produced from a population where steroids were being used for the sickest babies) we could randomize intubated babies at 14 days of age to a course of hydrocortisone between the STOP-BPD and NICHD trial schedules, and have restricted criteria for open-label steroid use, criteria which would be developed in order to predict mortality, rather than just remaining intubated. The primary outcome could be long-term pulmonary health, a hierarchical composite of in-hospital mortality, post-discharge respiratory mortality, readmission to hospital or ER visits for respiratory disease, and frequent respiratory symptoms. Construction of the long term health outcome should be a process involving all stake-holders, especially parents. I would be willing to ask parents to have their infants randomized in a study like that, would anyone be willing to fund it?
Without such data we we always be left prescribing these potentially toxic medications for short term gains but with unknown long term risks and benefits.
More generally, Bronchopulmonary Dysplasia should be considered to be an intermediate, surrogate outcome in trials, as an indicator of probable increased risk for long term respiratory morbidity, but not a firm end point in itself.








