The last time I blogged about this issue, there were a few comments from renal specialists which pointed out some limitations in my interpretation of the data. I defended myself admirably (it is my blog after all!) but I recognize that this is a complex issue.
Two new publications analyse serial serum creatinine in a cohort of preterm babies who all had multiple measures of creatinine concentrations. the first (Rios DR, et al. Creatinine filtration kinetics in critically Ill neonates. Pediatr Res. 2021;89(4):952-7) introduces the concept of creatinine filtration delay, with the idea that newborns have a delay before they start filtering creatinine of varying duration, being longest in the most immature infants. This is why creatinine concentrations take a while to fall and may indeed increase in the first few days of life.
I find this difficult to comprehend, if a baby is producing urine, then surely they must be filtering creatinine, even if GFR is very low (as it is immediately after birth) and tubular function is poor, I don’t think that there can be a selective delay in the filtration of creatinine. Perhaps they mean the term as a way of visualizing something else?
The idea I wrote about last time was that an increase in serum creatinine despite usual amounts of urine output may well be evidence of creatinine being reabsorbed in the tubules, one commenter noter that there is no reported mechanism for the reabsorption of creatinine, challenging that concept (that I must admit was not my idea, but the interpretation of a pediatric nephrologist of their data showing that creatinine clearance was lower than inulin clearance in the newborn rabbit, probably because of “back-leak of creatinine across leaky immature tubules“).
Around the time I posted that previous blog post, there was a publication from one of the commenters on the post (Askenazi D, et al. Acute changes in fluid status affect the incidence, associative clinical outcomes, and urine biomarker performance in premature infants with acute kidney injury. Pediatr Nephrol. 2016;31(5):843-51), which pointed out that infants lose extracellular fluid after birth, and the most immature babies lose the most weight after birth. This postnatal weight loss is due largely to a reduction in interstitial fluid, and thus to total body water. Creatinine is evenly distributed in all the body water compartments, therefore a reduction of total body water, without equivalent elimination of creatinine by the kidneys, will lead to an increase in serum creatinine concentrations.
I realize when re-examining the data from Rios et al, and their story of creatinine filtration delay, that they did not take into account the postnatal weight loss and, as the most immature babies have the highest extracellular fluid compartment (as much as 800 mL/kg, compared to about 400 at term), the impact of postnatal weight loss on serum creatinine concentrations is greatest among that group. I think it is most likely that the majority of the delay in the fall in serum creatinine is because of contraction of extracellular fluid and concentration of the creatinine while GFR is still low, rather than a delay in creatinine filtration.
I think this is an important part of the answer to the issues, Askenazi et al give a way of calculating an adjusted serum creatinine concentration accounting for the increased concentration due to weight loss. Which means, for example, that a small preterm baby who has a serum creatinine of 100 μmol/L at birth, the same as her mother, and then loses 13% of their body weight over the first 3 days and then has a re-measured serum creatinine of 120 μmol/L actually has a new adjusted serum creatinine of 100.5; If the baby weighs for example 1000g, and we assume a total body water, TBW, at birth of 800mL/kg then we can calculate the adjusted creatinine as 120 x (670/800) =100.5 (800 being the TBW in grams at birth and 670 being the TBW after loss of 130 g weight).
The total body creatinine, adjusted for weight loss, should be falling if renal function was “normal” to eventually arrive at a value which is appropriate for the size of the infant, but at least the adjustment for weight loss does give us a better indication of the real situation. Then we have to figure out why the adjusted serum creatinine is not falling, despite a urine output that has commenced.
This graph from Askenazi’s paper shows that this is a frequent finding in the very immature baby.
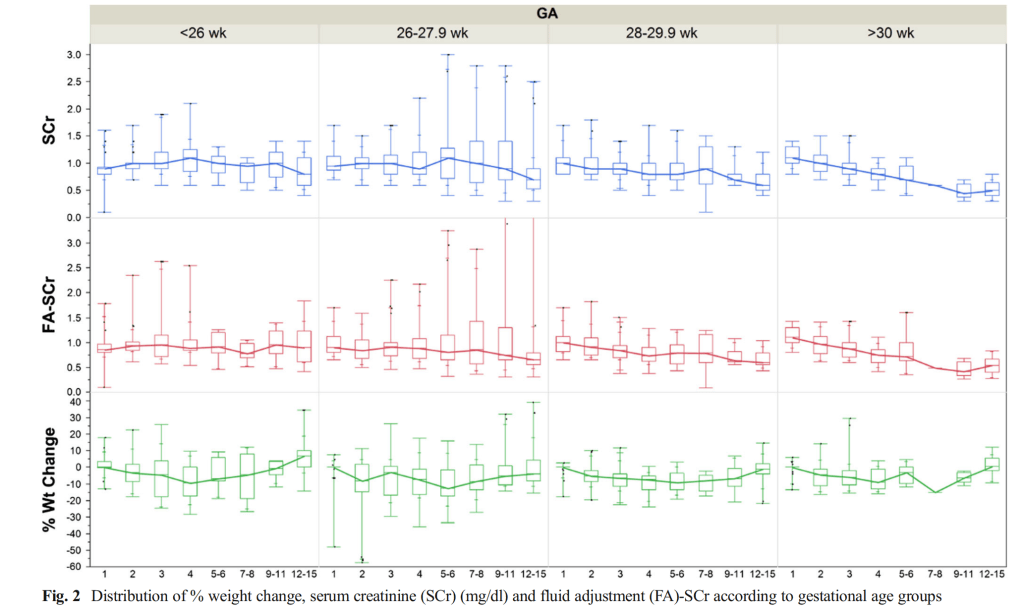
You can see that under 26 weeks, on average serum creatinine rises, but even the “fluid-adjusted” creatinine, taking into account weight loss, does not fall as you might expect; in more mature babies the “fluid adjusted” Serum Creatinine (FA-SCr) falls immediately after birth.
In this study there were 16 babies under 27 weeks of a total of 41 who had aute kidney injury according to the definition of an increase in serum creatinine of at least 27 micromol/L (0.3 mg/dl), 11 of them still had AKI by that definition when using adjusted creatinine values.
Of course, if there is glomerular filtration, creatinine is being filtered (I don’t think that creatinine filtration delay is a real thing) the Fluid-Adjusted Creatinine concentration will vary depending on the balance between GFR, and thus the creatinine clearance, and how much creatinine is being produced. Creatinine is produced by degradation of creatine, which is largely from muscle mass. So tiny preterm babies probably produce very little, but they also have very low GFR especially on day 1.
The other new study I mentioned is a study of 158 ELBW babies from Leuven, which modelled creatinine kinetics (van Donge T, et al. Characterizing dynamics of serum creatinine and creatinine clearance in extremely low birth weight neonates during the first 6 weeks of life. Pediatr Nephrol. 2021;36(3):649-59) and in their model they included a term for creatinine production which averaged 3.55 mg/day (SD 1.44) which is 31.4 μmol in modern units, the average weight of the babies was 820g, so a production of about 38 μmol/kg/d. (An adult produces somewhere around 160 μmol/kg/d).
These are the reference ranges they produced:
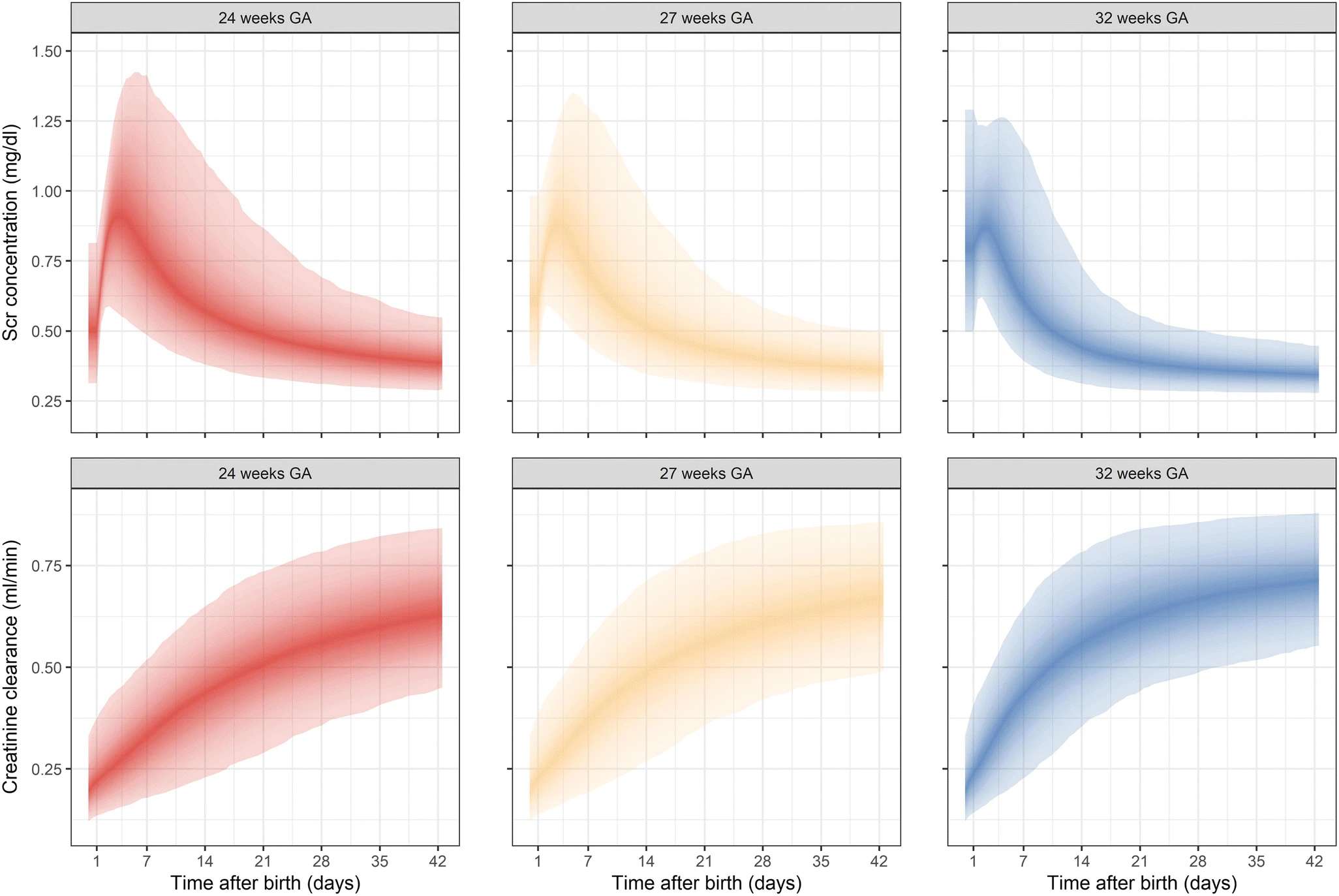
The model takes into account the fluid loss and reduction in total body water which leads to increased concentration in serum creatinine, the very low initial GFR, and the ongoing production of creatinine.
I think this is the best explanation of how all those factors interact, and gives reference ranges which can be used. It does use data from a group of babies of widely varying disease severity, some of whom were sick and at risk of kidney injury. However, their data did not seem to show an impact of inotropes, but did show a small impact of ibuprofen use.
How the use of these ranges helps in the definition of clinically important kidney injury will be important in the future. But it starts to explain to me the changes in serum creatinine in the very preterm.
For example: if creatinine clearance is as low as 0.2 mL/min on the first day of life, and an 820 g baby starts with a serum creatinine of 100 μmol/L, then they have a total body creatinine load of (0.820 x 0.8 (TBW) x 100)= 66 μmol. Filtering 0.2 mL/min means they filter 29 μmol of creatinine on day 1 while producing 31 μmol of creatinine, which would lead to an increase in total body creatinine load to 68 μmol, and if they lose 5% of their body weight (and 9% of their TBW) then their serum creatinine concentration will rise to 110 μmol/L.
A baby with a greater weight loss and a somewhat lower creatinine clearance will of course have a greater increase in their serum creatinine concentration..
I don’t know if you could use these data to try and back-calculate the creatinine clearance of an individual baby; then perhaps a certain lower limit of clearance could be used to define Acute Kidney Injury in the preterm. I do, however, think that these data clearly invalidate the definition of neonatal AKI as an increase in SCr of 27 μmol/L (0.3 mg/dL), and even using fluid adjusted SCr with that threshold. I think we need a new definition which is based around creatinine clearance which is lower than the gestational and postnatal age adjusted “normal”, or if that is too complex for routine use, then graphs such as those above with the addition of percentile lines may be enough for a working definition: a baby with a creatinine which rises too fast and crosses percentiles could be considered to have AKI.
I know I went a bit down the rabbit hole with this post, but I like understanding things, and when I don’t I get agitated and follow reference chains until I either get fed up or find a conclusion! I guess the take home message is that creatinine concentrations rise after birth, partially explained by postnatal weight loss, and also by low GFR, but we don’t have a good definition of AKI in the preterm infant.









Dr. Barrington – this is an excellent thread with a lot of critical thinking about a really important topic, thank you for writing about it!
I think many of us who study AKI in this population are starting to buy into what you have explained – perhaps the initial serum creatinine rise in the first 1-4 days in extremely preterm babies is not necessarily AKI for the reasons discussed above. However, I wouldn’t stop using the current definition of AKI in these babies beyond the first few days of age.
While we continue to search for the optimal neonatal definition of AKI – and it is possible that they will be based on curves with gestational age normals like you suggested, the rise in serum creatinine by 0.3 mL/dL after the first few days of age is an easy to recognize (and teach!) and important cut-off that has been associated with abnormal short and long term outcomes. In Dr. Askenazi’s recent paper (https://www.nature.com/articles/s41390-018-0249-8) evaluating optimal cut-offs in the AWAKEN population, they actually found the optimal SCr thresholds in the first week to predict AUC and specificity for mortality were ≥0.3 and ≥0.6 mg/dL for ≤29 weeks GA, and ≥0.1 and ≥0.3 mg/dL for >29 week GA. They concluded that unique SCr rise cutoffs for different GA improves outcome prediction and percent SCr rise does not add value to the neonatal AKI definition.
I don’t think that anyone who studies neonatal AKI will argue with you that a new and improved neonatal AKI definition is coming down the road and will hopefully include refined cutoffs and biomarkers of injury – but right now we shouldn’t dismiss the current definition, particularly after those first few days of SCr ‘rise’ in extremely preterm neonates. Thanks again for raising awareness about preterm neonatal kidney function and AKI!
Matthew Harer
After that initial rise, when the creatinine “should” be falling, then I would agree that it is likely that an absolute rise of 0.3 mg/dL or 27 micromol/L may well be a reasonable, and simple threshold. In fact, perhaps ANY rise during that period might be more sensitive and specific. Once the creatinine has stabilised and maternal creatinine has been filtered out, then probably a different threshold might be better, given the low muscle mass of our babies, and perhaps a smaller increase might be a better screening value.
The new article that you mention, (and that I would have referenced if I hadn’t already written too much) by Dr Askenazi, with Dr Ambalavanan as one of the authors, who wrote the other comment, is interesting, but my concern is that that study does not use Dr Askenazi’s own correction for weight loss, and many of the kids who have “AKI” using absolute rises in SCr during the first few days of life would not qualify using their own adjustment for weight loss.
I also question the relevance of the correlation between increases in serum creatinine and mortality, Its a long time since I saw a preterm baby die of renal failure, so I think the increase in serum creatinine might be a marker of serious illness but not necessarily a good marker of renal injury.
I agree with both you and Dr Ambalavanan that other biomarkers might well be interesting in the future, and more specific for renal injury.
Excellent post, as always! I think one issue of serum creatinine is that it changes late – by the time SCr increases even to a moderate extent, there is a substantial decrease in renal function (it’s more of Acute Kidney Failure rather than Acute Kidney Injury in the RIFLE classification [Risk–>Injury–>Failure–>Loss of Function–> End-stage]). I suspect that in the future there will be more sensitive biomarker-based methods for earlier detection of AKI at the injury phase (e.g. use of NGAL, Cys-C, KIM-1) when intervention may be possibly more effective (similar to use of troponins for MI).