MRI post-asphyxia, and post-rewarming, seems to be more predictive of long term outcomes than MRI at term for preterm infants. Imaging and analysis of the Apparent Diffusion Coefficient in the PLIC (posterior Limb of the Internal Capsule), for example, has reasonably good predictive value, and I like the fact that it is relatively objective, I can look at an MRI on a hospital computer, put my cursor on the PLIC and the software gives me a number; the lower the number the worse the outcome, in simple terms. I am not any good at interpreting MRIs by looking at the images, so getting a nice clear number appeals to me (and usually confirms what I think clinically). I can’t say it has ever really helped me in the clinical care of a baby.
We haven’t always been getting spectroscopy with our MRIs, but the new framework for practice from the British Association of Perinatal Medicine recommends the following
Where possible, Proton (1H) MRS Lactate/N acetyl aspartate (Lac/NAA) of the basal ganglia and thalamus should be performed with the MRI at 5-15 days after birth. This is the most accurate predictor of outcome in babies who have undergone TH therapeutic hypothermia.
The two references they give for that statement are 2 of the only three studies I am currently aware of. Mitra S, et al. Proton magnetic resonance spectroscopy lactate/N-acetylaspartate within 2 weeks of birth accurately predicts 2-year motor, cognitive and language outcomes in neonatal encephalopathy after therapeutic hypothermia. Arch Dis Child Fetal Neonatal Ed. 2019;104(4):F424-F32 and Lally PJ, et al. Magnetic resonance spectroscopy assessment of brain injury after moderate hypothermia in neonatal encephalopathy: a prospective multicentre cohort study. Lancet Neurol. 2019;18(1):35-45. The third one being Barta H, et al. Prognostic value of early, conventional proton magnetic resonance spectroscopy in cooled asphyxiated infants. BMC Pediatr. 2018;18(1):302.
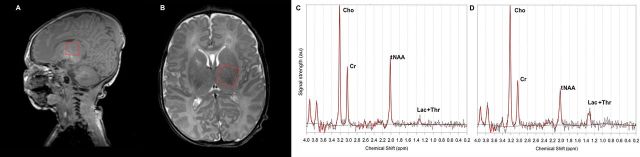
This figure shows basically what we are discussing, placing the voxel of interest in the thalamus, and then looking at the proton spectrum.
According to the framework for practice, the lactate to NAA ratio is highly predictive of adverse outcomes. Mitra et al (from where I took that image) refers to the Lac+Thr/tNAA (total NAA) which I think is the same thing as what Lally et al call the Lactate-NAA ratio. Barta et al also report that they calculated the Lac/NAA ratio, but that it was not one of the 3 ratios that adequately discriminated between the “good and poor outcome groups”.
The study by Lally defined an adverse outcome as death (I think there was only 1 death in this cohort) or a score on the Bayley 3 language or composite of <85 or cerebral palsy (GMFCS 2 or worse), seizure disorder or deafness, and has this figure :

Which shows a number of interesting things: firstly there are only 12 babies with an “adverse” outcome for the NAA concentration data (probably because it takes substantially longer to get this result, and perhaps because it is not available everywhere), there are about 26 adverse outcomes for the metabolite ratios; secondly, all of the measurements and calculated ratios show an overlap between the “normal” and “adverse” outcome babies. According to this study, the sensitivity of a Lactate-NAA ratio >0.22 is 88% and the specificity is 90% with the area under the ROC curve being 0.94. According to the same trial, an absolute NAA concentration <5.6 mmol/kg brain wet weight has a sensitivity of 100% and a specificity of 97% with an ROC curve area of 0.99.
If we look in detail at the results of Mitra et al, they included 55 infants with 16 deaths and 20 with an abnormal motor outcome, 19 with an abnormal cognitive outcome and 21 with abnormal language outcome by 2 years of age. By abnormal outcome they mean either death or a score on the Bayley3 composite of <85. They show this figure which I just can’t understand; if death or a low Bayley score on the cognitive composite was abnormal, then there should be 35 red dots, and 20 green dots, but there aren’t. There are actually more green dots than red, and even though it is difficult to count them there are more than 20 green dots.
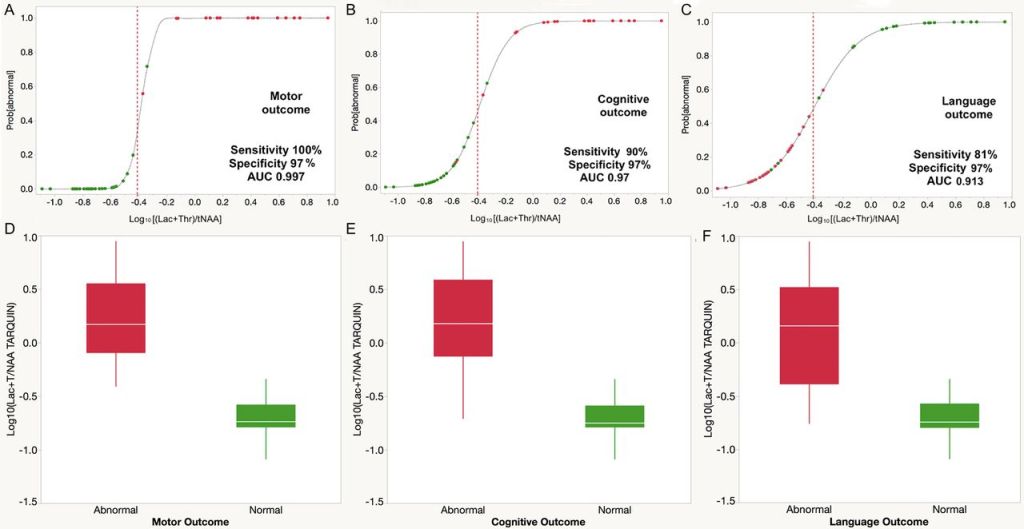
In addition, the dot colours are the wrong way round for the language outcome. More importantly, the cut off they use for determining what is abnormal is different to the threshold used by Lally et al, a Log10 of -0.4 is a Lac+Thr/tNAA ratio of about 0.4. The ratio used as the threshold value by Lally et all (0.22) gives a Log10 value of -0.65, which, looking at these figures, if you used the threshold that Lally et al used for these babies in the Mitra study, it would classify many of their normal babies in the abnormal group.
Looking at these data I really do not see that there is enough data to support performing MR spectroscopy on all babies with HIE.
- We can with some degree of confidence, based on about 45 babies, state that if the Lac/NAA ratio is low, then outcomes are probably going to be worse than if they are higher. But what threshold ratio we should use to make this determination is not clear, and it doesn’t seem to work in Hungary.
- Does it really matter if a Bayley 3 score is <85? I think that dividing babies up into the “normal” and “abnormal” is unhelpful, and creating a category that includes 16% of the non-asphyxiated population and calling them “abnormal” is questionable. Dichotomizing the richness of human development should be avoided.
- What is the clinical value to an individual or their family that is added by performing MR spectroscopy? Does knowing the Lac/NAA ratio help the family in some way? Does it help them to access services, or prepare for the future, or is there some other benefit?
- Does MR spectroscopy adequately differentiate between the babies with very severely abnormal outcomes (such as an inability, or very restricted ability, to communicate) and babies who have no, minor, or moderate disability?
As far as I can see currently, perhaps spectroscopy can help us to perform asphyxia intervention studies with shorter follow up. If we can confirm that interventions that have no impact on spectroscopy have little or no clinical benefit, then MR spectroscopy as a biomarker for research might be very valuable to screen out things that are unlikely to improve outcomes.
The absolute NAA concentration, based on a very small sample, might be a better biomarker, but it currently takes an extra 25 minutes in the magnet, if you have the right 3T magnet and the software.
As for clinical use as a routine after HIE, the data regarding NAA concentration or the metabolite ratios are based on very small numbers of babies, with outcomes that are not very important.
If future studies can focus on extremely adversely affected babies, and if they use the same thresholds to classify studies as abnormal or not, then we might in the future have enough data to support using routine spectroscopy as a clinical tool.








