The above title is the title of a talk I just gave at the NEO2019 conference. I have made available a .ppt file of the final slides from the talk, under the tab at the top of the page “presentations”. The version in the App, which is made available to participants at the conference, is slightly different to this final version.
My review of the literature led to the following conclusions
Among very preterm or very low birth weight infants :
Growth and bone mineralization approaching desired standards can only be achieved by fortifying BM
Commercial bovine or human multicomponent fortifers have become the standard of care
Desired growth can be achieved with maternal BM and fortification, or donor BM and fortification, if enough attention is paid to growth
Donor BM has less protein (and slightly fewer calories) than preterm maternal BM, for a few weeks, and requires higher supplementation
And then in terms of the scientific evidence about what and when:
When maternal breast milk supply is insufficient to meet the baby’s needs: supplementation with formula increases NEC compared to donor BM (Older and recent studies, moderate to good quality data)
Multi-component fortification, with powdered bovine-protein based products, has not been shown to affect NEC compared to no fortification (almost all studies before 2000, poor to moderate quality data, wide confidence intervals)
Multi-component fortification from different sources (bovine compared to human) has not been shown to change the incidence of NEC, when used with a strategy of maternal or donor breast milk (moderate quality data, wide confidence intervals)
Individualized fortification using BM analysis not shown to improve clinically important outcomes compared to adjustment according to growth (poor quality data, small studies, wide confidence intervals)
Early introduction of fortifiers has not been shown to adversely impact clinical outcomes or complications compared to >100 mL/kg/d (poor quality data, one study, wide confidence intervals)
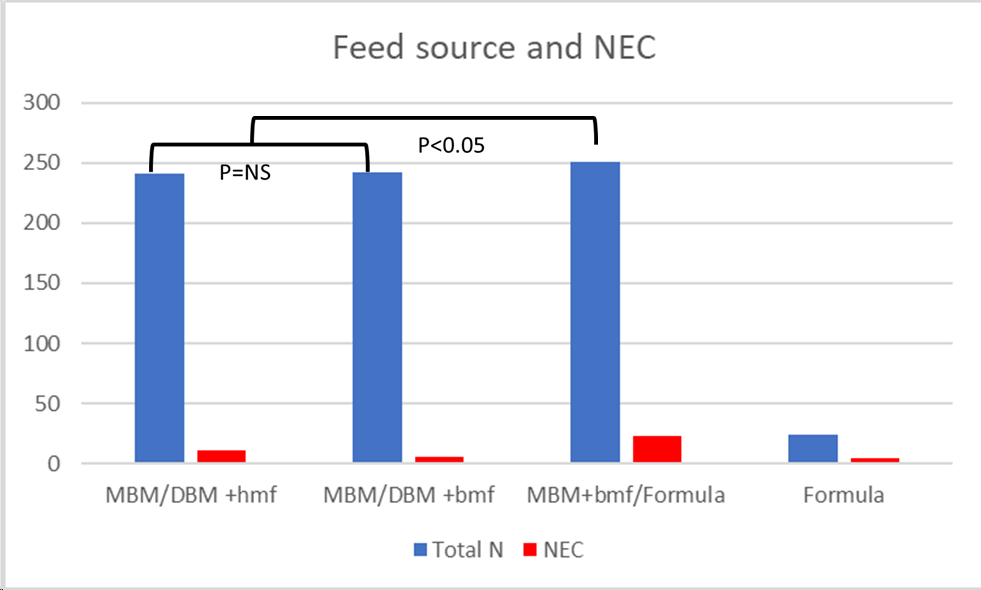
The data about human versus bovine based fortifiers for expressed breast milk feeding in the very preterm are based on only 3 small studies. All of which I have discussed previously on this blog. I created a graphic using the data from these trials, comparing the overall incidence of stage 2 NEC or greater with each feeding strategy. The strategies being 1. Maternal BM with Donor BM as a supplement when insufficient MBM, both fortified with human-milk-based fortifier (MBM/DBM+hmf) 2. Maternal BM with Donor BM fortified with bovine-milk-based fortifier (MBM/DBM+bmf) 3. Maternal BM, fortified with bovine-milk-based fortifier with preterm formula as as supplement. 4. Preterm formula.
Cristofalo EA, et al. Randomized trial of exclusive human milk versus preterm formula diets in extremely premature infants. The Journal of pediatrics. 2013;163(6):1592-5 e1. Sullivan S, et al. An Exclusively Human Milk-Based Diet Is Associated with a Lower Rate of Necrotizing Enterocolitis than a Diet of Human Milk and Bovine Milk-Based Products. The Journal of pediatrics. 2010;156(4):562-7.e1. Trang S, et al. Cost-Effectiveness of Supplemental Donor Milk Versus Formula for Very Low Birth Weight Infants. Pediatrics. 2018;141(3).O’Connor DL, et al. Effect of Supplemental Donor Human Milk Compared With Preterm Formula on Neurodevelopment of Very Low-Birth-Weight Infants at 18 Months: A Randomized Clinical Trial. JAMA. 2016;316(18):1897-905.
Strategy 1 was an intervention group as each of the first 3 of those trials, Strategy 2 in Trang and O’Connor, strategy 3 in Trang and Sullivan, and strategy 4 in Cristofalo.
I stress that this is not a formal SR and meta-analysis! The p=values are the typical p=values from the initial publications
I think it is interesting to compare the tiny amount of information we have about different sources of fortifiers and milks for their impact on NEC, to the evidence which exists from RCTs of probiotics. The latest systematic review/meta-analysis included about 3,600 patients per group.
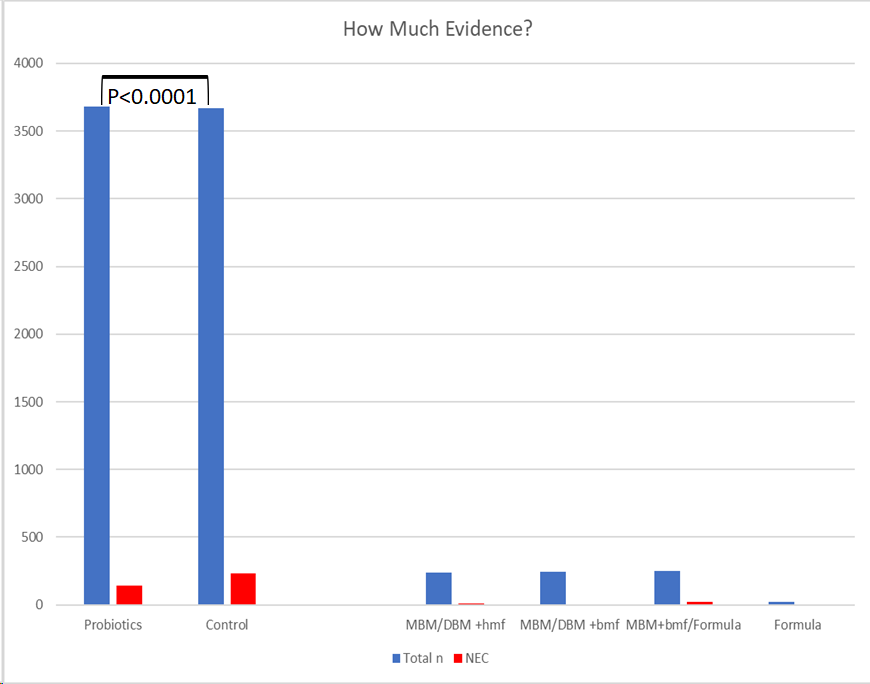
I find it distressing that fortifiers, which are given to fragile, high-risk, babies 8 (or 12) times a day for several weeks, do not have to provide the same kind of proof of safety and efficacy as a new drug, which may be given 2 or 3 times a day for a week. We end up in a situation where thousands of babies are being exposed to these agents with very poor quality evidence that they are equivalent to each other.
Given these limitations of the proofs, an evidence-based protocol for breast milk fortification would look like this
For infants at risk of NEC:
Promote maternal breast milk as much as possible, early expression, lactation consultants, pumps freely available everywhere…
When MBM insufficient, always use donor BM, until risk of NEC passed (no good data on when to stop donor milk, 34 weeks post-menstrual age seems reasonable)
As feeds advance, fortify breast milk as soon as TPN can not meet nutritional requirements of the infant (which will usually happen around 50 mL/kg/d)
Start with standard fortification, up to an assumed calorie density of 24 kcal/oz for maternal BM, and start at a higher dose for donor BM (assumed calorie density of 26 kcal/oz) because donor BM has less protein.
Use powder or liquid fortifier, there is no proven advantage of one over the other.
Use bovine or human-based fortifier, there is no proven advantage of one over the other if MBM is supplemented with donor BM.
Concentrate on growth, review frequently and increase fortification if growth < target for 2 wk, at ≥ 160 mL/kg/d, then re-assess frequently.









Can you post the talk please ?