My first foray into systematic review and meta-analysis was about 25 years ago with this article Barrington KJ. The adverse neuro-developmental effects of postnatal steroids in the preterm infant: a systematic review of RCTs. BMC Pediatr. 2001;1:1. As you can see it was the very first article in the new journal BMC Pediatrics, I had submitted to BioMed Central, BMC, because it was a new open access model of publishing, (which authors didn’t have to pay for!) and then they asked me if I minded them transferring it to BMC Pediatrics.
At that time there was evidence that infants exposed to dexamethasone had an increase in the frequency of Cerebral Palsy. Unfortunately, in the RCTs, many of the control infants received dexamethasone. Which makes evaluation of the impacts of the steroid in the long term very difficult, if not just about impossible. This was the Forest plot of the impacts of steroids (all dexamethasone except for the study by Fitzhardinge et al, which was a mega-dose of hydrocortisone).
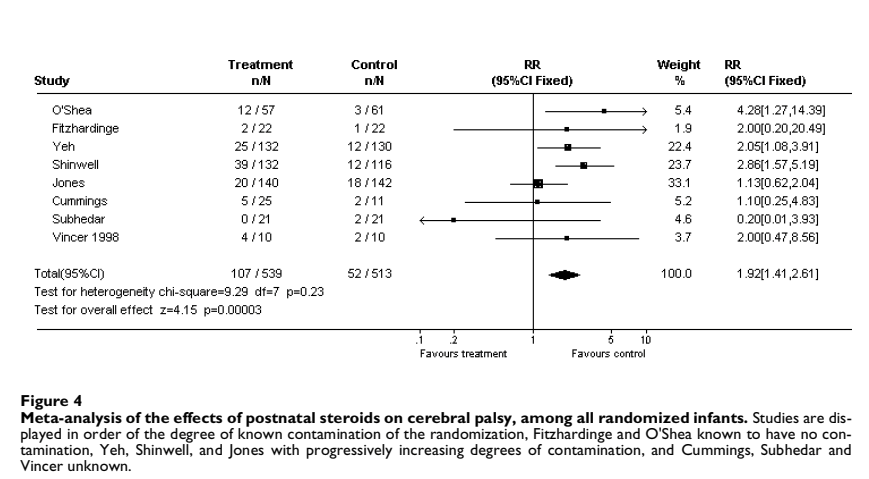
As noted in the figure legend, the study by O’Shea et al, which was a 42 day course of dexamethasone starting on day 15-25, was the only modestly sized trial without any crossover, control babies were not allowed to receive postnatal steroids.
Unfortunately, since then we have very unreliable data, because we can’t help ourselves, and frequently treat the babies randomized to control with steroids. In the trials in my SR/MA, Shinwell’s trial, 33% of surviving controls received dexamethasone, and in the Jones trial, 40% of the surviving controls received at least one course of dexamethasone.
This also means that we don’t know what the long term impacts of postnatal steroids are on the lungs! It is very disappointing, that so few of these trials have had long term pulmonary outcomes reported. If we are giving steroids to improve respiratory health in the long term, surely measuring that would have been reasonable? One of the few trials to measure long term pulmonary outcomes was that Jones trial, with its 40% contamination rate. They followed the subjects out to 13 to 17 years of age. And found no benefit of postnatal steroids at any point in their follow up.
I don’t really care if a baby is still in oxygen at 36 weeks. And neither do their parents!
Do postnatal steroids decrease the number of babies whose respiratory health adversely impacts their life? Or their family? Needing oxygen at home, needing respiratory interventions at home, re-admission to hospital, frequent cough or wheeze, impacts on feeding or on physical activity, recurrent stridor…. Those are things that bother parents, and we have no idea if postnatal steroids improve any of them. (Thivierge E, et al. Pulmonary important outcomes after extremely preterm birth: parental perspectives. Acta Paediatr. 2023;112(5):970–6).
I’m coming back to this issue because of a new ST/MA which has been published. (Raghuveer TS, et al. Systemic steroids and bronchopulmonary dysplasia: a systematic review and meta-analysis. J Perinatol. 2026). The search strategy found 44 studies. Their analysis found no change in mortality after adjusting for steroid contamination, and no change if the analysis was limited to the 24 trials with no steroid contamination. I was surprised that they were able to find 24 trials with no steroid treatment of the controls, and indeed I was right to be surprised.
Some of those trials did in fact have contamination of the controls with steroid treatment. Yeh 1999, for example, had several babies, 12% of controls, who received steroids after the first 28 days. Among the studies that report neurological outcomes, only O’Shea et al (which they call Kothadia, the first author of the initial publication), among the trials with data on CP, was not contaminated by steroid use in the controls. There also seem some errors with the actual results, in Stark et al table 2 reports 12 placebo and 8 dexamethasone babies with moderate to severe CP, and the text adds that 4 placebo and 5 dexa babies had mild CP, for a total of 16 vs 13 cases; the Forest plot in the appendix has only 6 vs 8 cases of CP. Also Yeh’s study, in table 4, reported 12 placebo and 25 dexamethasone babies with CP, while this new Forest plot has 17 vs 9. I’m sure they read the original publications carefully, so I don’t understand where those discrepancies come from.
However, the biggest problems with the outcome data continue to be the very small numbers of infants followed, and the use of steroids among controls. In Stark’s NICHD trial, for example, 50% of control babies received dexamethasone. It is impossible to say anything reliable about the long term impacts on CP or development, or pulmonary outcomes with this extremely serious limitation. When it comes to long term toxicities, analysis by Intention to treat is no longer the only analysis that should be performed. An analysis by treatment actually received, should have been performed. It seems likely that steroids given early in postnatal life, might have more marked effects on neurodevelopmental outcomes, and that bigger doses will have greater impacts. Meta-regression examining the impacts of those factors would be very useful, especially if the dose of dexamethasone actually received was available, and entered into the analysis.
I can find such evidence in any published article that shows an improvement in long term respiratory health with postnatal steroid use compared to untreated controls.
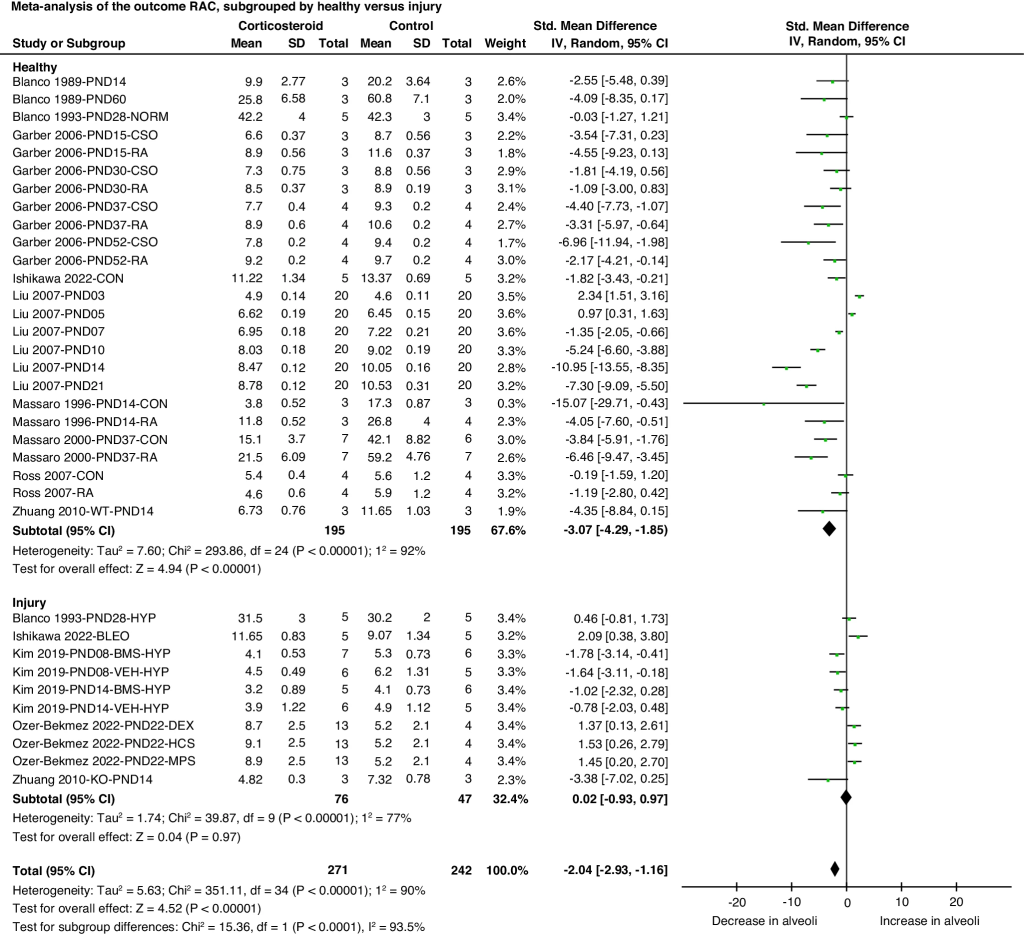
This is vitally important. Steroids reduce inflammation and thus the number of babies who need oxygen at 36 weeks. But there are known adverse impacts of systemic steroids on lung growth and development. I don’t remember ever quoting previously a systematic review of animal studies on this blog, but this one is great! Lok IM, et al. Effects of postnatal corticosteroids on lung development in newborn animals. A systematic review. Pediatr Res. (96) 2024. p. 1141–52. Despite the limitations of the initial studies, the differences in methodologies and in outcome measures, making meta-analysis very limited, and for some measures, impossible, it is quite clear there are serious negative effects of steroids in immature mammals on alveolarization, the radial alveolar count, for example, a well-validated measure of alveolar development, is substantially decreased by postnatal steroid use, but, it seems only in healthy animals, not after a lung injury (of various types).
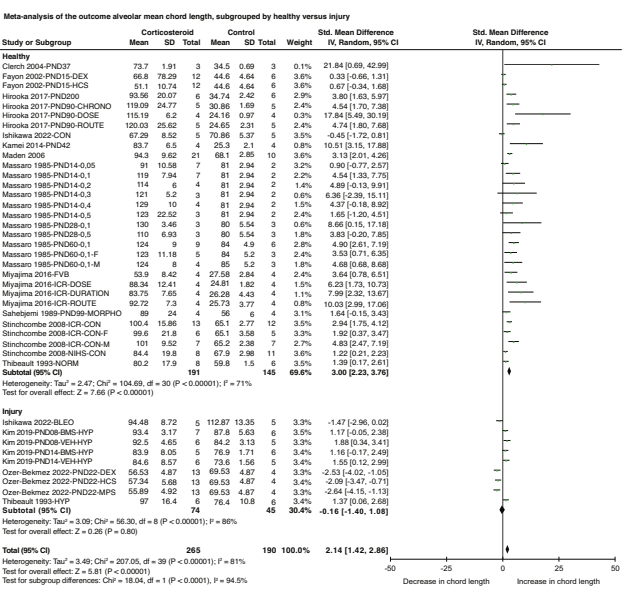
Another measure of alveolarization, that I didn’t know about, is the mean alveolar chord length, which increases when there are fewer alveoli, That measure also shows substantial adverse effects of postnatal steroids in animals (mostly rodents) with normal lungs, but less evidence of such toxicity if they are given after a lung injury,
The conclusion of this review is
Postnatal corticosteroids have a negative effect on body weight and lung development resulting in persistent alveolar simplification
Although postnatal steroids reduce “BPD” there is no evidence that they improve important clinical features of lung injury. The animal data in fact suggest a major risk of adverse impacts on lung growth and alveolarisation.
Moderately large RCTs of dexamethasone with ≤ 33% treatment of controls with steroids (O’Shea, Yeh and Shinwell) have all shown an increase in CP with the steroid treatment. All of the larger RCTs performed since my 25 year old SR/MA have been contaminated by 40% or more of treatment of controls with steroids (which is basically just Stark and Doyle). Its really disappointing, looking back, that even though there was a serious concern raised so many years ago, none of the more recent trials have seen fit to analyze cerebral palsy as an outcome, and none have reported CP according to dose actually received.
This latest meta-analysis blithely reports that the steroids do not show evidence of increased risk of “NDI”, and focuses on a reduction in oxygen needs at 36 weeks. Wouldn’t it be great to know if we are actually helping our patients when we prescribe steroids for them?
Unfortunately the data we have do not show any improvement in pulmonary health with postnatal steroids, and there remains a major concern about the potential impacts on motor development and cerebral palsy.









Thank you for yet another good post.
From what I remember of the discussions on this matter on old Twitter, it was said that early Dexametasone = possibly worse outcome and Cerebral Palsy, while Dexametasone closer to term or in high risk for severe BPD could be favourable. So rather than just no for all, maybe we should say yes to some?
Do you have any comment on that? Or of the use of hydrocortisone presented in this recent Swedish study: https://pubmed.ncbi.nlm.nih.gov/41712209/
Among the many meta-analyses that have been published, yes there is some evidence that earlier use of postnatal steroids is more neurotoxic. Also if the risk of death is higher, the relative benefits seem greater. But even then, the long term pulmonary benefits are uncertain, of course that becomes less important if there is a difference in survival.
As for the Swedish study, that really addressed early prophylaxis, the Premiloc approach. Dose of hydrocortisone is much lower than treatment doses. That cohort analysis showed less “BPD” but slightly worse mortality. I would love to know if there was a difference in oxygen at 40 weeks, which is more important to families, or in pulmonary outcomes in the long term. Again, if it is just a reduction in inflammation, which leads to less oxygen need at 36 weeks, but without a long term benefit, then the increase in mortality, and in late-onset sepsis is more important. The dose is probably higher than the “physiologic” dose, (whatever that means for a baby born at 25 weeks) so it could possibly have adverse effects on lung growth and alveolarisation.
Thank you for your thoughtful insights on these important issues. Please allow me to make some additional comments. The highlighted conclusion quoted from the Lok manuscript (PMID 38493255) regarding glucocorticoids and alveolar development is itself at risk of oversimplification. Most importantly, it does not include the important distinction between the glucocorticoid effects on lungs with vs without underlying inflammatory injury. When glucocorticoids were given in the context of lung injury in these animal studies, there were no observed negative effects on alveolar count, lung surface area, or mean alveolar chord length (Table 2). At the same time, nor was there evidence that glucocorticoids overcome the negative consequences of the underlying inflammatory injury on alveolarization. In the setting of evolving BPD, there have been no reported negative long term pulmonary outcomes in randomized studies of glucocorticoid use. There are, however, at least some findings supporting pulmonary benefit (as measured by FEV1) in follow-up at 8-11 years (Nixon, 2007, PMID 1382108) or (in a study comparing two dexamethasone regimens) at 15 years (Gross, 2005, PMID 15741372).
The evidence from studies in animals with otherwise normal lungs is indeed compelling that postnatal glucocorticoids can impair alveolarization. The direct application of these findings to humans is less clear (see commentary PMID 38914764). The alveolar phase in rodents is only a few weeks (primarily postnatal days ~4-21). In humans, alveolarization commences at ~36w gestation and continues for many years into childhood. Even a short course of steroids thus spans a much longer percent of lung development in rodents compared with humans. Perhaps this is among reasons for the lack of identified negative pulmonary consequences of even prolonged courses of high dose glucocorticoids administered during the alveolar phase in humans, e.g., as historically used in infants to treat hemangiomas (PMID: 15313812).
Regarding the risk for cerebral palsy with early dexamethasone use, data reported in the study by Yeh et al (PMID 9310536) may provide some important unappreciated insights. The mean pCO2 was significantly lower in the group treated with dexamethasone (at days 4, 6, and 21). One standard deviation below the mean pCO2 at each of these timepoints was ~24-25 mmHg in the dexamethasone group and ~30-32 mmHg in the control group. This would place a greater number of infants in the dexamethasone group at risk for hypocarbia-associated white matter injury and related motor deficits. Whether similar consequences of dexamethasone on ventilation occurred in other early-use studies is unknown. It is worth bearing in mind that these studies were designed to assess dexamethasone use as a preventative measure rather than a form of treatment, and acute effects on lung mechanics may not have been anticipated. Moreover, they were performed at a time when the potential risk for cerebral palsy with prolonged moderate hypocarbia was unknown.
As with any medical treatment, individualized risk:benefit assessments best inform use of glucocorticoids in managing the inflammatory component of lung disease in preterm neonates. And as with every medical treatment, the understanding of these risks and benefits evolves over time.