I had a long break from blogging over the summer, and, as I return to action, I note that there are some really high quality studies which have been performed or are in progress. But I continue to lament some of the primary outcomes chosen. I am not alone in being very concerned that the classical neonatal outcomes, which combine death with another outcome of either clinical importance, or sometimes of questionable clinical significance, are so ingrained in our research design practice that they are difficult to alter.
As one example, a really important study, by a researcher, and research group, for which I have enormous respect, has just published their 2 year outcomes (Dargaville PA, et al. Two-Year Outcomes After Minimally Invasive Surfactant Therapy in Preterm Infants: Follow-Up of the OPTIMIST-A Randomized Clinical Trial. JAMA. 2023). Of course, you can guess, the primary outcome of the long term follow up was “death or NDI”!
Often this outcome is chosen for 2 reasons, 1. they are competing outcomes, in the sense that if you are dead you can’t have developmental delay, and 2. it improves the power of the study.
Both of these reasons are flawed.
- There are many other competing outcomes, and there are many ways of analyzing them that take into account their relative importance. The Win Ratio, that I have written about a few times, is one such, but other methods exist. It is possible, therefore, to analyze the effects on survival, and other “competing” outcomes, without treating them as equivalent.
- Power is only increased if the outcomes vary in the same direction. And indeed, the reason for using these composite outcomes is sometimes because of concern that they might change in different directions! If they do, then power is reduced.
As an example of this, the primary outcome of the early stage of this trial (Dargaville PA, et al. Effect of Minimally Invasive Surfactant Therapy vs Sham Treatment on Death or Bronchopulmonary Dysplasia in Preterm Infants With Respiratory Distress Syndrome: The OPTIMIST-A Randomized Clinical Trial. JAMA. 2021;326(24):2478-87) was, if you remember, “Death or BPD”, and there was a small increase in death, and a larger decrease in “BPD”, which led to, overall, the results of the trial being considered “null”, that is, there was no significant difference in the primary outcome. There were 485 infants randomized, and the results showed the following:
Death or BPD occurred in 105 infants (43.6%) in the MIST group and 121 (49.6%) in the control group (risk difference [RD], -6.3% [95% CI, -14.2% to 1.6%]; relative risk [RR], 0.87 [95% CI, 0.74 to 1.03]; P = .10).
Some journals would probably not have allowed the details that followed to be in the abstract, details which show that mortality was 2% higher, but BPD was 8% lower with MIST compared to standard care.
Incidence of death before 36 weeks’ postmenstrual age did not differ significantly between groups (24 [10.0%] in MIST vs 19 [7.8%] in control; RD, 2.1% [95% CI, -3.6% to 7.8%]; RR, 1.27 [95% CI, 0.63 to 2.57]; P = .51), but incidence of BPD in survivors to 36 weeks’ postmenstrual age was lower in the MIST group (81/217 [37.3%] vs 102/225 [45.3%] in the control group; RD, -7.8% [95% CI, -14.9% to -0.7%]; RR, 0.83 [95% CI, 0.70 to 0.98]; P = .03). Serious adverse events occurred in 10.3% of infants in the MIST group and 11.1% in the control group.
This is to me a good example of why not to use such composite outcomes. I don’t know what a win ratio type analysis (or another way of prioritising a composite) would have shown, but the published analysis implies that death and needing oxygen at 36 weeks are equivalent, and that MIST had no effect.
The new publication of the 2 year outcomes reports, as the primary outcome, “death or NDI”, (they actually call it “NDD” or neurodevelopmental disability, in this trial) which triggers the same kind of reflection as previously. Because of the variety of sites involved in the trial, and the variety of follow up therefore available, the definitions of so-called NDD are different to anythong previously published, and are as shown in the table below, from the online supplement. The “PARCA-R” is a parent questionnaire, the abbreviated questionnaire was an alternative 6-question instrument, used when the PARCA-R was not available.
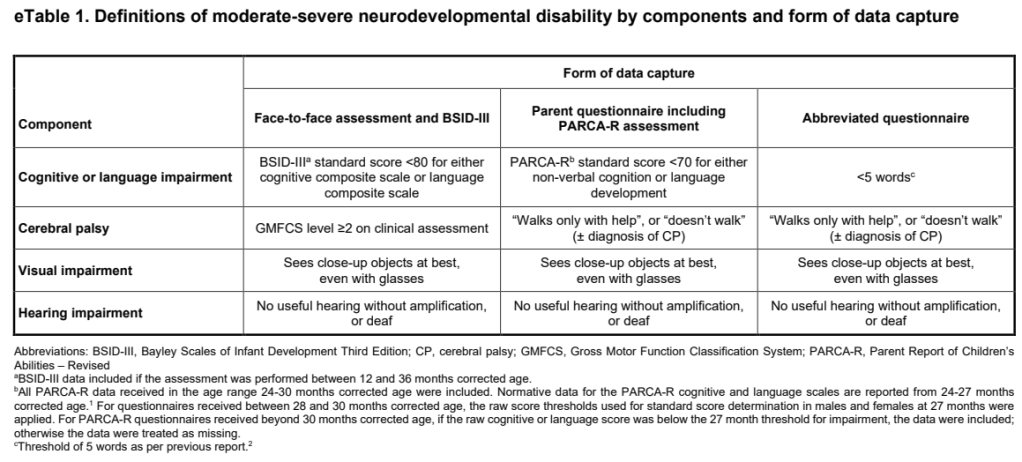
Did anyone think that being dead and having one of these characteristics were equivalent bad outcomes? I also wonder why use a measure of neuro-development as the primary outcome for the trial at all, I certainly didn’t think it likely that MIST, even if beneficial for respiratory outcomes, would have an impact on developmental outcomes. And I have to repeat another of my pet peeves, why refer to these outcomes, low scores on the BSID cognitive or language composite, or PARCA-R scores <70 as a “cognitive impairment”? I must insist that such low scores are not impairments. An impairment is “Any loss or abnormality of psychological, physiological, or anatomical structure or function” according to the WHO definition. Slow language development does not qualify.
Despite all these concerns, I think surveillance of developmental outcomes in a study like this is important, just to make sure that there is nothing unexpected. And, indeed there were no differences in the various domains of neurological abnormalities or developmental progress between MIST and control groups.
Important long term respiratory outcomes are vital for studies such as OPTIMIST, and they are described in this new publication.
There were another 5 deaths in each group between 36 weeks PMA (the deaths reported previously) and 2 years of age, so by 2 years of age, 12.9% of the MIST babies, and 10.5% of the controls had died.
I am not sure who developed the list of outcomes, and whether parents were involved in choosing them, but the reported respiratory outcomes probably all had a measurable negative impact on the baby and/or the family.
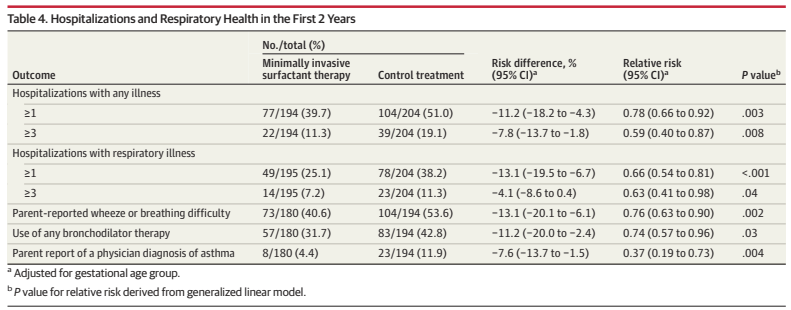
As you can see, these aspects of respiratory health, as reported at 2 years of age, are all better in the MIST group than the controls. To recall, the study babies were between 25 and 29 weeks gestation, were on non-invasive respiratory support with >30% oxygen in the first 6 hours of life. Babies in the control group had a sham MIST procedure and then returned to CPAP or NIPPV, and in both groups they were intubated for surfactant if they subsequently needed more than 45% oxygen (or >40% and a worried neonatologist!). There was a dramatic reduction in the proportion of babies needing intubation in the first 3 days, from 72% to 37%, with the MIST approach.
At a recent meeting I heard some comments about the value of the comparison in this study, but I don’t think we should criticise the authors for not having done a different trial! These long term results confirm that MIST/LISA at 30% oxygen is better than being intubated for surfactant at 45%, in terms of lung injury. Is MIST at 30% preferable to INSURE at 30%? Or preferable to MIST at 45%? This study cannot answer those questions.
Of note, infants in the trial were not permitted premedication, other than sucrose and/or atropine for the MIST procedure. Laryngoscopy is painful and unpleasant and leads to hypertension, bradycardia, desaturation and increased intra-cranial pressure. I hate the idea of non-premedicated laryngoscopy, but I don’t know what approach is the best, we need good trials of premedication for MIST/LISA. Finding a cocktail which will allow continuous spontaneous respiration, while decreasing the pain and adverse physiologic changes should be a priority.








