The Finnegan score was developed and evaluated in the 1970’s as the first systematic way of monitoring babies going through perinatal drug withdrawal. Although it was an advance at the time, it was not developed using modern standards, and was never, I don’t believe, studied in a randomized trial, indeed it would be hard to know what you could have compared it to, other approaches were rather haphazard.
The Finnegan scoring system, and the treatment approaches based on it, became the default approach, and have been the standard of care for many years. The problem of neonatal drug withdrawal seems to be getting much more frequent, especially in the USA, reliable prevalence data are of course difficult to obtain, but 7% of US mothers report taking opioids during pregnancy, 1/5 of them report abusing opioids, rather than therapeutic use. The incidence of neonatal abstinence syndrome, NAS, nearly doubled between 2000 and 2017, and I think all indications are that it is still increasing, with 7 cases per 1000 births in 2017. I.E. nearly 1% of all births in the US.
In Canada in 2020 the incidence was about 6 per 1000 live births (not including Quebec who collect data separately). The opioid crisis in Canada is, generally speaking, worse in the west of the country and is spreading eastward, with the highest rates of overdose deaths in British Columbia, followed by Alberta then the prairies and Ontario, with far fewer cases as yet in Quebec.
With this enormous and increasing incidence, evidence-based methods for evaluating and intervening for these infants were required, and a new program, first reported in 2017 was developed, based on a simplified evaluation (which is where ESC comes from: is the infant able to eat, to sleep, and to be consoled), use of non-pharmacologic measures to calm the infant, reduced stimulation, skin to skin care, encouraging breast feeding, and limiting morphine use.
Several publications strongly suggested that the ESC approach was followed by a reduction in duration of hospital stays, and less opiate administration. This new publication (Young LW, et al. Eat, Sleep, Console Approach or Usual Care for Neonatal Opioid Withdrawal. N Engl J Med. 2023) reports the first large RCT, performed as a cluster randomized trial, with all the 26 centres switching from standard care to ESC, but doing so at times which were determined randomly; a “stepped wedge” design.
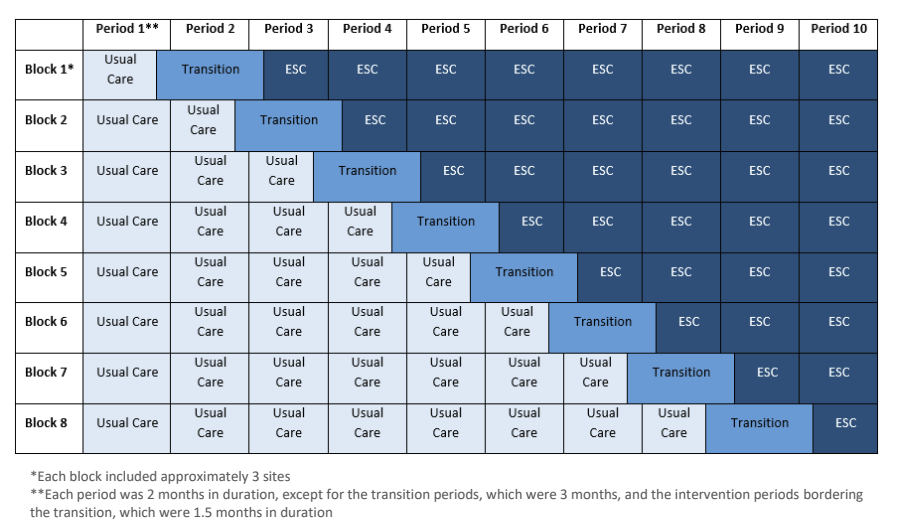
It would be easy to criticise this design, it is almost implicit in the design that ESC is preferable! No cluster was randomized to switch from ESC to standard care, which you could do if there was really equipoise. I think it shows that most people were already convinced that ESC was the better approach, but they wanted some scientifically valid way of confirming that. The design does give real-world information about the size of the impact of ESC, and allowed some post-discharge evaluation of safety.
The primary outcome of the trial was the age of being medically ready for discharge, defined as : “an age of at least 96 hours, a period of at least 48 hours without receipt of an opioid, at least 24 hours with no respiratory support and with 100% oral feeding, and at least 24 hours from initiation of maximum caloric density.” over 1300 babies were enrolled, and 837 were discharged when medically ready, and therefore contributed to the primary outcome.
There were therefore 468 who were discharged before they met this definition, almost all were either <96 hours of age (n=211), or it was <48 hours since their last opioid dose (n=231). The investigators therefore also evaluated a “modified definition of medical readiness for discharge” which included an age of at least 72 hours and at least 24 hours without receipt of an opioid. This definition could be evaluated for 1164 of the infants.
Just getting the babies home earlier, while a benefit of itself, is not necessarily an advantage if the babies need to be re-admitted for further NAS therapy, or for other related reasons, or if parental coping is affected and more babies have adverse outcomes. The babies were therefore followed for 3 months post-discharge for the following outcomes “(any acute or urgent care visit, emergency department visit, or hospital readmission), and a composite critical safety outcome at discharge and through 3 months of age (nonaccidental trauma or death).”
Follow-up of these babies may be very difficult, especially in the US health environment. “Outcomes after hospital discharge were assessed prospectively at 3 months of age by means of a review of electronic medical records (including linked medical records) and media review through a search of public records (e.g., news reports, obituaries, and registries)”.
The main results are below: as I mentioned, the primary outcome was assessed for 837 babies, but the actual mean length of hospital stay includes all the babies, as does the dramatic difference in the proportion who received morphine.
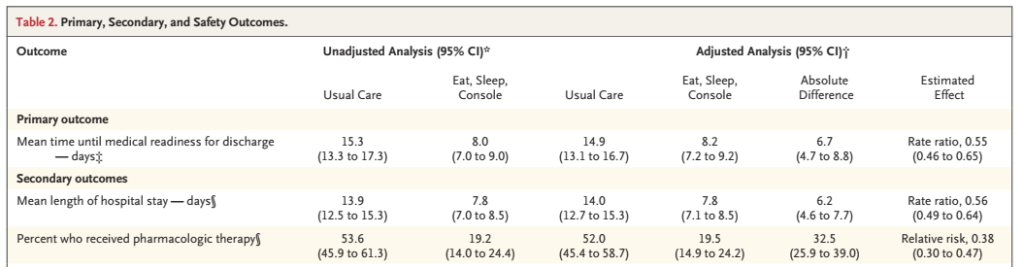
The modified definition occurred at 14.5 days in the usual care group, and 8,1 days in the ESC group.
For the 3 month outcomes :
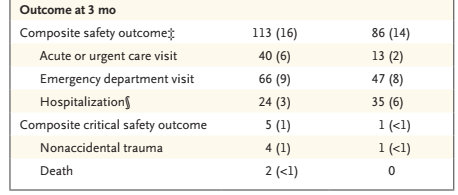
About 2/3 of the hospitalisations in each group were possibly related to NAS.
This study should be the death-knell (whatever a “knell” is) for the Finnegan score. It is not particularly objective, nor clearly very useful for determining therapy. The simplified ESC system should rapidly become the standard of care, and the standard against which any innovations in NAS care should be tested.
As one example, there are several trials of buprenorphine for NAS, as an alternative to either morphine or methadone, those trials (such as this one) have generally used the Finnegan system, and have often shown shorter treatment with buprenoprhine than the alternative. The effect size of ESC is larger than that shown in those trials, in general. Maybe some of them should be redone, with ESC as the approach to care in both groups.








