As many of you will know, I have advocated for many years for evaluation and management of very preterm babies based on their tissue perfusion rather than the blood pressure. There is little or no correlation between BP and perfusion, and many of the commonest interventions, in particular dopamine, may increase BP almost exclusively by vasoconstriction, thereby worsening tissue perfusion. Such an approach, however, suffers from the difficulty in evaluating tissue perfusion in the preterm infant, clinical signs are limited, and lab tests may be misleading. Echocardiography is relatively objective, but difficult, has some inter-rater variability, and multiple repeated studies are often needed during management, leading to major disturbance of fragile babies.
A reliable continuous method with minimal invasiveness would be a major step forward. The perfusion index of some pulse oximeters excited me a few years ago, and a study that my group performed showed a useful correlation of perfusion index in limited circumstances, at 6 hours of age among babies with a low SVC flow.
A group from Saitama in Japan have been studying laser doppler skin blood flow for a few years. They have described the normal transitional changes in flow, and noted that low flow may precede later intraventricular haemorrhage. In contrast to my statement above, they looked at the impact of dopamine on skin blood flow, and showed that dopamine appeared to increase both blood pressure and skin blood flow, at least blood flow in the lower limbs (increased by 5%), blood flow to the skin and subcutaneous tissue on the forehead was not affected, despite a 10% increase in mean blood pressure; I don’t know of any other study that shows an increase in systemic perfusion with dopamine in compromised preterm babies, but even I must admit that the currently available data are poor. If mean BP increased by 10%, but perfusion locally increased by between 0 and 5% that is some evidence of vasoconstriction, but perhaps locally in some vascular beds there may be a sufficient increase in perfusion pressure to improve perfusion.
The big question with any such method, is whether using the method can improve management to the extent that outcome are better. We know that, statistically, babies with lower blood pressure have worse outcomes and that babies who receive treatment have worse outcomes than those who do not; which means that treatment is either a risk factor itself, or is a marker of increased risk. We tried to differentiate between those possibilities in the HIP trial, but were unable to enrol enough babies to find a definitive answer.
The Saitama group have just published the results of an RCT to try and answer the question, does treatment according to the results of the laser doppler device improve clinical outcomes? (Ishiguro A, et al. Randomized Trial of Perfusion-Based Circulatory Management in Very Low Birth Weight Infants. J Pediatr. 2021). They randomized VLBW infants to have one of 2 approaches, one based on normalising the blood pressure, and the other on normalising the blood flow measure.
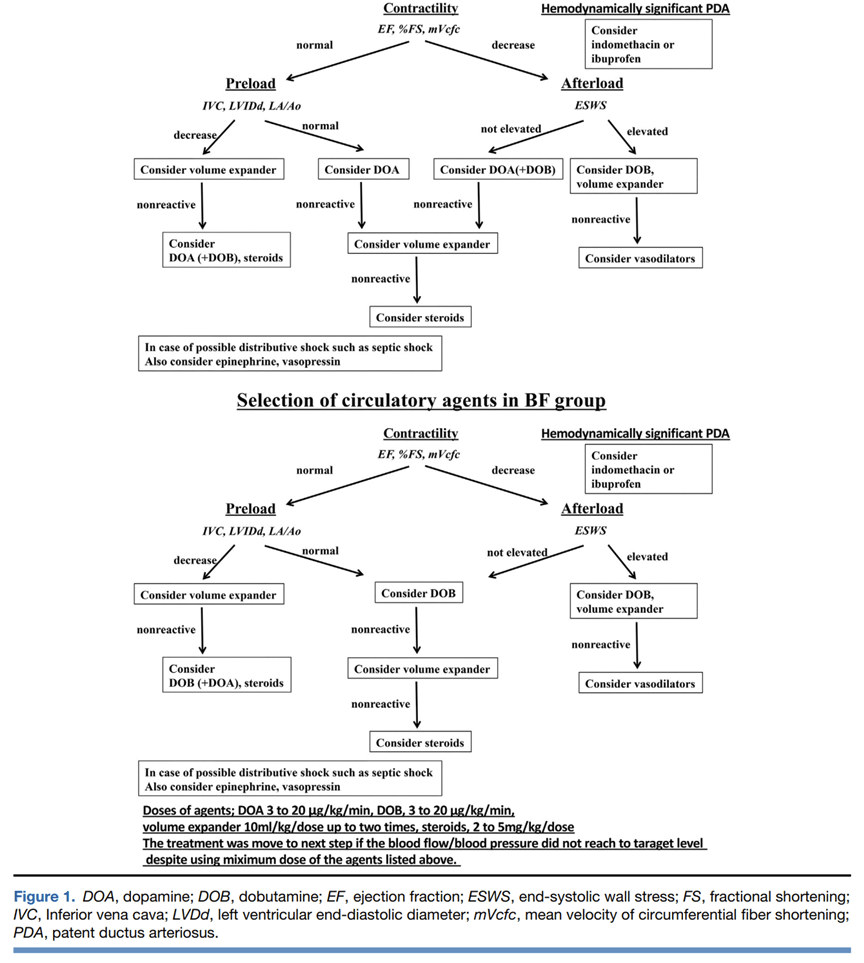
The 440 VLBW infants were randomized to one of 2 contrasting protocols within 6 hours of birth, if they had no IVH on screening head ultrasound. Not all the infants had invasive blood pressure monitoring which is a reasonable pragmatic choice (and is, in fact, what I wanted to do for the HIP trial), but immediately introduces uncertainty, as non-invasive blood pressures are somewhat unreliable, especially in the smallest babies, and especially if the baby is hypotensive. All babies had skin blood flow measured from one foot, and were treated by the assigned protocol if the BP was below the gestational age in weeks, or if the BF (blood flow) was below 14 mL/min in the first 24 hours or 17 mL/min thereafter; depending on group. If they passed their treatment threshold then a functional echocardiogram was performed (in addition to a routine daily echo) and treatment given according to the schema above. The primary outcome was any intraventricular haemorrhage. They also evaluated how often the treatment was successful in improving the randomized parameter to above threshold within 3 hours.
The trial was a single centre study that took 7 years to complete, randomizing about 2/3 of eligible admissions. 37% of the BF group and 42% of the BP group passed their treatment threshold and had intervention. In the BF group they almost all had dobutamine, and nearly half of those also had dopamine. In the BP group about half of those with intervention received dopamine, and a similar proportion received dobutamine. In both groups 12% had a volume bolus and 6% had hydrocortisone.
I would question some of the reasoning that went into constructing these algorithms, but overall, I think they are not unreasonable. On the other hand, I find it a bit surprising that one in five VLBW infants had low blood flow with normal contractility with decreased preload indices and didn’t respond to a fluid bolus (as that is the only way a BF baby could get dopamine added to their dobutamine); which makes me wonder how well the algorithms were actually followed. Indeed I find it a bit surprising that such a high proportion of VLBW infants were considered to need cardiovascular support, they were enrolled early in life and for 40% of the babies to need some cardiovascular intervention in the first 72 hours seems aggressive. They do however have a historically low proportion of babies with IVH, in the CNN about 30% of VLBW babies have at least a grade 1 IVH, small subependymal haemorrhages being included in their outcome of IVH. Overall the outcomes are the typically very good outcomes of Japanese centres with a remarkably low mortality, with 1 death out of 440 VLBW babies within the first 7 days, and only 7 deaths prior to discharge, very low NEC and low late-onset sepsis are also evident.
The sample size was calculated based on that previous incidence of IVH of 13%, with a power to detect a reduction to 6%. I presume, though it is isn’t mentioned, that the previous IVH rate was in the context of BP-directed management of the infants. They actually showed an 11% rate of IVH in the BP group, and a 7% incidence in the BF babies. The 95% compatibility limits for the difference in IVH rate (not given in the manuscript) are a 9.4% absolute risk decrease in IVH and a 1.2% increase.
The study included some babies at relatively low risk, so the infants of 1001-1500 g birth weight only had 2% vs 3% IVH rate. It would be just about impossible to design a trial to find a reduction in an adverse outcome from 2%. Among the ELBW babies there were 18% IVH in the BP babies, and 10% with BF targeting, which give 95% compatibility limits of -17% and +0.5% absolute risk difference.
The intervention was not always successful at increasing the target parameter within 3 hours of initiating treatment, and a very interesting post-hoc analysis is presented, which shows that the BP and BF group babies who never received intervention had very low rates of IVH, (3% vs 6%) but those who had a successful increase in their BF did better than those who had a failed application of the protocol (i.e. BF stayed low at 3 hours of intervention) whereas the low BP babies who continued to have a BP below GA at 3 hours of age did better than those in whom BP increased.
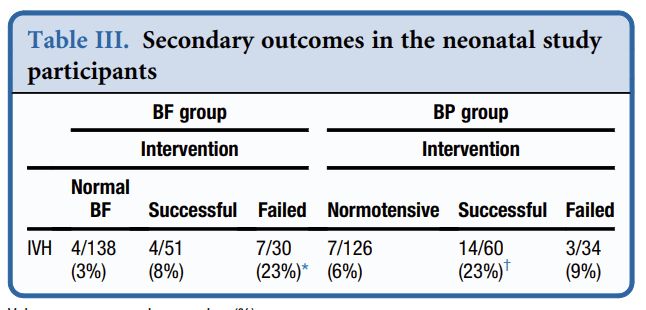
This obviously has to be interpreted with a great deal of care, but does suggest several possible interpretations.
This study helps in calculating sample sizes for future trials, 11 of the 39 IVHs occurred in babies who had no cardiovascular intervention. I don’t have any way of calculating how many of those babies had both normal BF and normal BP throughout the trial, but a baby who stayed above both thresholds throughout 72 would have had no change in management. If we guess that about 40-50% of babies will have no cardiovascular intervention regardless of which group they are in, and perhaps 25% of IVH occurs in those babies, then that impacts the numbers of babies required for a trial to show that intervention algorithm reduces IVH frequency. It is complicated, of course, because some babies with low BP will have normal flow, and vice versa.
I think this trial is clearly not enough evidence for a universal change in practice, but it is strong evidence that we need to do more investigations of this way of managing cardiovascular support in the newborn. Refinement of the protocols, comparison with other indicators of perfusion (perhaps cerebral NIRS, perhaps perfusion index) and a larger sample size, or perhaps concentrating on the babies at highest risk, are needed to answer the vitally important questions posed by this study.









I wonder how well toe temperature would correlate with bloodflow measured this way
There are a few studies looking at toe temperature, or at least the difference between core and skin temperatures, they suggest that it might be a very useful early marker of late-onset sepsis.
I’m not sure how useful it would be as a measure of systemic perfusion, but is probably worth further investigation, especially as it doesn’t take any new equipment.