I was one of the investigators in the PINT trial (Kirpalani H, et al. The Premature Infants in Need of Transfusion (PINT) study: a randomized, controlled trial of a restrictive (low) versus liberal (high) transfusion threshold for extremely low birth weight infants. J Pediatr. 2006;149(3):301-7. that we published 14 years ago. That RCT found little difference in any clinical outcomes between higher and lower transfusion thresholds among 450 preterm babies with a birthweight less than 1kg. Of note, there was no difference in NEC or other typical neonatal complications. The follow-up portion of that trial showed no difference in the primary outcome, but there was a hint on post hoc analysis that perhaps the Bayley version 2 MDI was less likely to be below 85 in the liberal transfusion group.
The only other sizable RCT in the literature, until now, enrolled 100 babies < 1300g birthweight, and again showed very little impact (Bell EF, et al. Randomized trial of liberal versus restrictive guidelines for red blood cell transfusion in preterm infants. Pediatrics. 2005;115(6):1685-91). That study was reported as showing a possible increase in the combined outcome of grade 4 IVH and PVL with restricted transfusion guidelines, but if you look at the results grade 3 haemorrhages were much more frequent in their liberal transfusion group (16% vs 2%) and if you examine all the significant brain injury together, there was no difference between groups.
Hence the need for more trials. At least 2 large multicentre RCTs have been performed, and the first to be reported is ETTNO (Franz AR, et al. Effects of Liberal vs Restrictive Transfusion Thresholds on Survival and Neurocognitive Outcomes in Extremely Low-Birth-Weight Infants: The ETTNO Randomized Clinical Trial. JAMA 2020;324(6):560-70). In this trial 1,000 babies < 1kg birthweight were randomized at less than 72 hours of age to receive transfusions when their hematocrit fell below either higher or a lower threshold. The thresholds were adjusted by postnatal age and whether the babies were “critically ill” or not. The schedule is here, and I am also including the notes, which describe the criteria for being “critically ill”.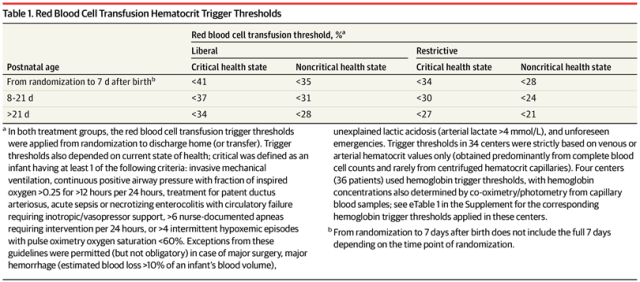
If you are not used to thinking in haematocrit, just divide by 0.3 to get the approximate haemoglobin.
In general, I think this schedule is reasonable for an RCT; to be doable, a large multicentre trial needs to have methods to which enough people will buy in, and to be relevant, the criteria have to be applicable in future everyday practice, even if you are not sure that some of it makes sense. You could certainly criticize why babies in more than 25% oxygen on CPAP need to have a higher threshold than babies in 21%; the concentration of oxygen being inspired has no impact on tissue oxygen delivery. Also, higher thresholds for babies with frequent apnea or frequent hypoxic episodes could be questioned.
The primary outcome of the trial was “death or neurodevelopmental disability”, you can probably already hear me screaming! (Softly, into my coffee). The neurodevelopmental part of that is also referred to as “cognitive deficit”. The developmental part of the outcome, which is, as usual, the biggest contributor to the outcome, is based on a Bayley version 2 MDI score of <85 at 24 months corrected age. It is, of course, ridiculous to refer to a low score on a developmental screening test as an “impairment” or a “deficit”, especially when 1 SD is chosen as the cut-off, using this terminology 16% of all human babies are “impaired” or have “a deficit”. Combing a lowish score on a developmental screening test with death as the primary outcome is really lumping together 2 things that should clearly be evaluated separately. Neither of the previous trials showed any tendency to impact mortality, the primary outcome of this trial could easily have been developmental delay or neurologic impairment at 24 months corrected among surviving babies. Combing 2 outcomes of such disparate importance risks diluting the real importance of a trial, if mortality had by chance been slightly higher in one group than the other, then a difference in the important long term outcomes could easily be hidden, or all sorts of other possibilities arise. I don’t think there is any a prior reason why both mortality and developmental progress at 24 months should be expected to change in the same direction by this intervention. Mortality is so much more important than having a low-ish Bayley score that the risk of missing an impact on survival by including it in a composite with a much more common outcome is really quesitonable.
I think this would have been a good trial to analyze survival separately, and then to examine long term impacts on development and on neurologic dysfunction. But; to stick with the analysis plan of the published primary outcome, there was no apparent difference between groups. 
More importantly, as survival was about identical, (91% restricted vs 91.7% liberal) and there was less than 10% loss to follow-up, we can have a great deal of confidence in the developmental outcomes which were close to identical between the groups. Mean Bayley 2 MDI scores 92.5 in each group with very similar numbers below -1SD and below -2SD, cerebral palsy and PDI scores similarly were not different between groups, and the more rare components of neurologic dysfunction (visual and hearing impairment) were also very similar.
As for shorter term outcomes they are listed in this table
You can see that there isn’t really a hint of a difference between the groups for any outcome. Also not in this table, but in another, the duration of respiratory support was identical, the age that caffeine was stopped was the same.
I think the complete lack of impact on NEC is a good sign that transfusion-associated NEC is a mirage. Twice as many low threshold babies as high threshold babies never had a transfusion (40% vs 21%), overall they received double the blood volume. The supplemental data show the average hematocrits in the two groups which are not enormously different, with few values being below 27, but there are large differences in the number of late transfusions between groups. NEC was also similar between groups in the two previous RCTs that I mentioned at the start of this post.
One concern about this otherwise excellent trial is that 25% of babies had already received at least one transfusion with packed red cells before randomization. That seems like an awful lot of transfusions in the first 72 hours, I guess that, being enrolled between 2011 and 2014, there were probably a lot of babies that did not get a placental transfusion, but it still seems like a high proportion of early transfusions in these infants.
The implications for clinical practice are that either of the transfusion schedules, or something in between them, are appropriate for extremely low birth weight babies and will not likely have an impact on short or long term outcomes. To make it simple (I like to make things simple) critically ill babies who have a risk of limited cardiac output responses to anaemia could have a threshold for transfusion throughout their hospital stay of a haematocrit of 34 (haemoglobin of 110 g/L) and stable babies the threshold could be 28 (haemoglobin of 90 g/L) in the first week of life, falling to a haemoglobin of 80 in week 2 and 3, and 70 thereafter.










Excellent summary thank you
very good blog post!
Yes, it’s indeed an excellent interpretation of the studies presented. Additionally, being a practicing Neo for nearly 3 decades I’d please like to broach the highly pertinent questions regarding anemia and practice of neonatology. Many of these issues have been adequately addressed in the future but we might have forgotten about them completely. Here are the issues:
1. Is a low hct level associated with apnea of prematurity?
2. Is a low hct associated with a decreased cellular oxygen extraction?
It’s my understanding the answer’s to both’s NO
However, unfortunately, in neonatal practice we tend to be preoccupied with correcting some specific target or arbitrary number without proof of any beneficence and often not even considering the potential maleficence. I remember, nearly 30 years ago, in many NICUs, there used to be a standing order to check the Hct of all the babies in the convalescent unit and just tank them up with blood to attain the targeted number. I’m highly delighted to see the marked reduction of the total number of blood transfusions prior to discharge. We’re also slowly appreciating the complex compensatory physiologic factors determining oxygen extraction. The accurate clinical parameters that can ascertain global oxygen sufficiency are yet to established. Just like oxygen, we’re slowly realizing that a higher hct is not necessarily more beneficial!
Pingback: Do blood transfusions treat apnoea of prematurity? | Neonatal Research