I received a comment from Colin Morley (everyone in neonatology knows who Colin is, for anyone else who is reading this, he is a neonatologist and researcher from Cambridge and Melbourne). I asked his permission to post it (with my reply which is below) and he kindly agreed:
My take on this is that at the moment it is a bit like saying antibiotics are good although there we have lots of data about individual antibiotics and specific infections.
The problems with probiotics are:
- There are no well proven probiotic combinations available for prescription in the UK or Australia. I dont know about Canada.
- We dont know which probiotic or combination to use.
- We dont know the dose to give.
- We dont know when to start.
- We dont know when to finish.
- Should they be given in the same way to breast milk fed or formula fed babies.
- Although it may appear there are no infections with the probiotics this is possibly because they dont grow in standard blood culture media.
So I think we need more detailed specific studies to help us answer the above problems. I would not suggest people go to the local high street pharmacy and buy probiotics for their prems because they often do not contain what they say on the tin or in the dose stated.
Hopefully the Australian ProPrems trial data (1200 babies) will be available soon. Even with that number it has little power to show a believable difference in NEC so it is targeting infection, which is a nightmare, what is infection?
My response was as follows:
In Canada we do have preparations with good quality control approved by Health Canada (not specifically for preterms, but at least approved to be sold with an official number), made in approved factories etc.
My response to most of your other comments are that none of the questions will be answered by the ongoing trials. We need comparative trials.
I think the situation is a bit like hypothermia a few years ago, we knew it worked but the AAP refused to endorse treatment because we didn’t know how long to cool for or exactly what temperature. We still don’t know those things, but the widespread introduction of cooling was delayed and many babies suffered as a result.
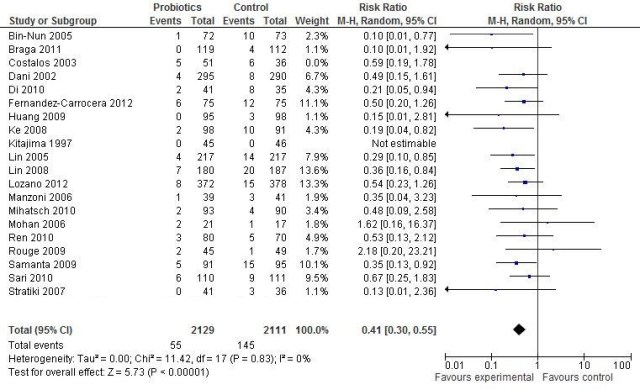
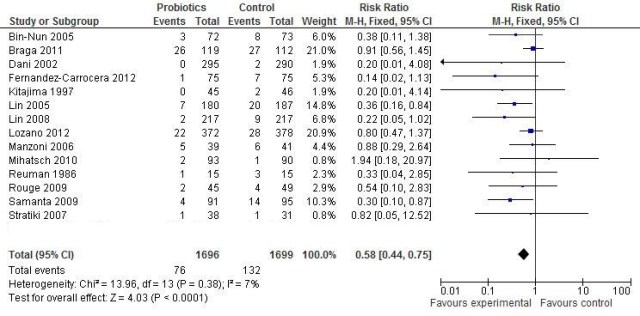
It really doesn’t seem to matter which organism is used. In fact the intestine is colonized with different profiles of organisms around the world, as long as you have either a lactobacillus or a bifidobacterium in the stuff the effects seem to be about the same. But a streptococcus thermophiles or a saccharomyces doesn’t work (there are now 3 studies of saccharomyces, 1 published and 2 presented, none show an effect).
I agree that the difficulty culturing the bugs is a problem, but, even if there are some undetected infections, mortality is still reduced. The reported infections have been relatively minor and easy to treat.
The unreliability of mixtures bought in the health food stores in the UK and the USA is a huge problem for using them. A reliable preparation is essential.
Colin then replied further:
I have one more comment to your comments. Although comparative studies are theoretically a good idea they are going to be almost impossible if NEC is the outcome. If the rate of NEC is 7% then even an unlikely halving down to 3.5% is going to need thousands of babies.
What is needed is a good study with large numbers (many of the probiotic studies are too small and may be publication bias), a reasonable incidence of NEC say 7% (some of the trials have very high levels in the controls – 13.6%, 16.6%, 22.8% etc and so are probably not representative of what happens in most neonatal units) and a probiotic combination which has been shown to be consistent in composition and in number of bugs and is licensed by the national bodies like the FDA and Australian TGA. If we have that then we can be comfortable to proceed and treat all babies.
When I was in Taiwan I asked them what they did. I was told if the parents want probiotics they have to buy it from the local pharmacy!
I agree with the statement that we need reliable preparations. We are fortunate in Canada to have the Natural Product Authorization system.
It is certainly true that the sample sizes for comparative trials will be huge. There is an international group that has formed who are interested in setting up a registry for the cohort studies that are being done, with a long term goal to perform some comparative RCTs which will probably be cluster randomizations, to account for cross-colonization within an NICU. I think we need to set up networks to do studies such as those, with a simple data set to keep it manageable and less expensive.