There are a few new publications which might help us to answer the question posed in the title of this post.
When heated, humidified, high flow nasal cannulae were first being spoken about I remembered an old study using standard cannulae but with flow rates up to 2 litres per minute. In 13 infants with an average body weight of about 1500g, the authors measured intra-oesophageal pressures, using either 0.2 or 0.3 cm diameter prongs. (Locke RG, et al. Inadvertent administration of positive end-distending pressure during nasal cannula flow. Pediatrics. 1993;91(1):135-8).
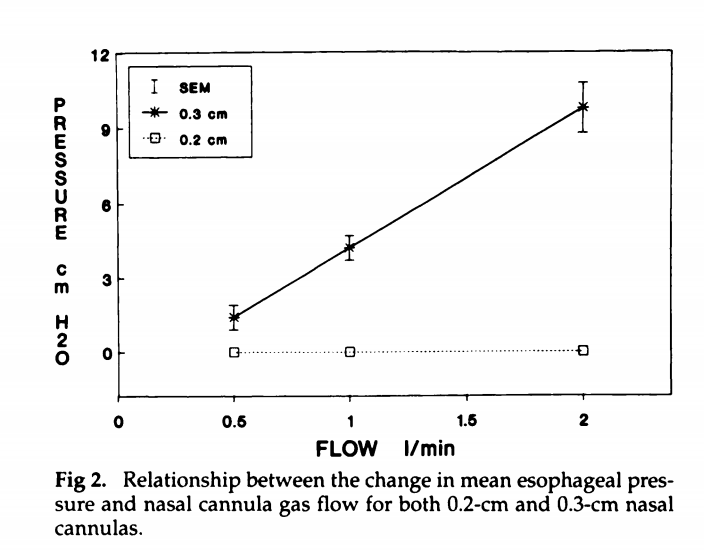
As you can see from this figure you can get very high oesophageal pressures with 2 litres per minute of flow if you have tight-fitting nasal prongs. The average was 9.8 cmH2O, and the error bars are SEM, which means the SD was 3.6, so the upper limit of pressures generated could easily be 9.8+2SD= 17cmH2O! The lower limit would be about 2 cmH2O with a 2 litre flow, and as you can see a pressure of 0 when the smaller prongs were used. You can see from the title of the article that the PEEP delivered, at that time, was considered “inadvertent”!
Since then the use of heated humidified gases and systems specifically designed for high flow have been introduced. Unfortunately, the initial introduction of HFNC was without good trials evaluating risks and benefits. I, for one, was reticent to use them for quite a while. The only advantage I could see was that they seemed to be more comfortable for larger, more mature, kids, although the trials evaluating pain scales are contradictory. There does, however, seem to be less nasal trauma when they are used for younger preterm infants (<28 wk PMA), and parents generally prefer them, perhaps because it is easier to interact and play with their babies with HFNC than CPAP.
We started using them for older babies with mild to moderate BPD who still needed CPAP when they were getting to be over 34 weeks. One of the features of our unit is that babies on HFNC can be transferred to the intermediate care section of the unit, whereas if on CPAP they have to stay in an intensive cot. When we were short of beds we sometimes switched a baby from CPAP to HFNC to make room for a new admission. So the use of HFNC gradually crept up, which made us review our practices and ask a couple of questions based on more recent data than the article from 1993.
In terms of the impact of HFNC on the respiratory system, they do generate PEEP under certain circumstances, but it is variable from baby to baby, and from minute to minute. It depends on how tight they fit in the nostrils of the baby. and the flow rate and whether the mouth is open or not.
And here I can’t help myself, but I must insist: NARE IS NOT A WORD! Sometimes the nostrils are referred to as the “nares” (pronounced naireez) which is a Latin word occasionally used in English to refer to the two nostrils. One nostril in Latin is “naris”, so, if you wanted to, you could refer to a naris, but I would insist that the whole sentence is in Latin! Quid magnum naris! (what a big nostril!) We have a perfectly functional English word for the nostril, let’s use it!
As another aside, one thing I find cute in a French-speaking NICU is that nasal flaring is referred to as “flapping the wings of the nose” (battement des ailes du nez).
To return to medicine…
A recent article on the physiological impacts of HFNC has, for the first time in preterm babies, I believe, tried to actually measure one of the supposed mechanisms of action, that is, dead space washout. (Liew Z, et al. Physiological effects of high-flow nasal cannula therapy in preterm infants. Arch Dis Child Fetal Neonatal Ed. 2020;105(1):87-93) They did this in 44 low birth weight babies (500 to 1900g), testing different flow rates and comparing to nCPAP at 6 cmH2O.
Normally, when you inspire, you at first pull into your gas exchange sacs (terminal sacs or alveoli depending on GA) the gas that you just expired, from the tracheobronchial tree then the upper airways, before getting fresh atmospheric gas (there is, of course, a gradual mixing during inspiration). The idea of dead space washout with HFNC is that a high flow of gas into the pharynx, much higher than the infant’s minute ventilation, will wash out the pharynx with a fresh gas flow (21% oxygen or more and 0% CO2) and thus decrease the effective dead space. To measure this you could look at the moment by moment gas composition of the gas in the pharynx during the respiratory cycle, and determine the inspiratory concentrations of CO2 and O2. This group did almost exactly that, but taking into account the mixing of gases and the turbulence caused by the HFNC in the pharynx, they decided to measure the peak, end-tidal CO2. Which dropped progressively as gas flow increased.
As you can see the pEECO2, or end-tidal CO2, expressed as a percentage dropped from 2.3% at a flow of 2 to 0.9% at 8 litres per minute, confirming that there was indeed a wash-out of the dead space. In addition, minute ventilation fell, although not as consistently, as flows increased, which is what you would expect; if you wash out CO2, then PCO2 will fall, leading to a decrease in respiratory drive and then in minute ventilation until PCO2 comes back up to where it was, this is confirmed by the stability of the transcutaneous CO2 (about 46 mmHg in old units). This might mean that a baby who has a high work of breathing associated with BPD will have a reduction in their respiratory effort as flows increase, which is in fact what we sometimes see in the NICU.
As another aside, babies (and indeed adults) with chronically raised CO2 have intact CO2 responses. This has clearly been shown in adults with COPD who do NOT become “dependent on hypoxic drive” as stated in some texts, which has led in the past to restricted oxygen administration. Newborns also remain responsive to CO2, even when chronically hypercapnic. I studied this back when I was a fellow, in the last century, giving 2% CO2 to a few babies with BPD and chronic hypercapnia, they increased their minute ventilation, just as you would expect.
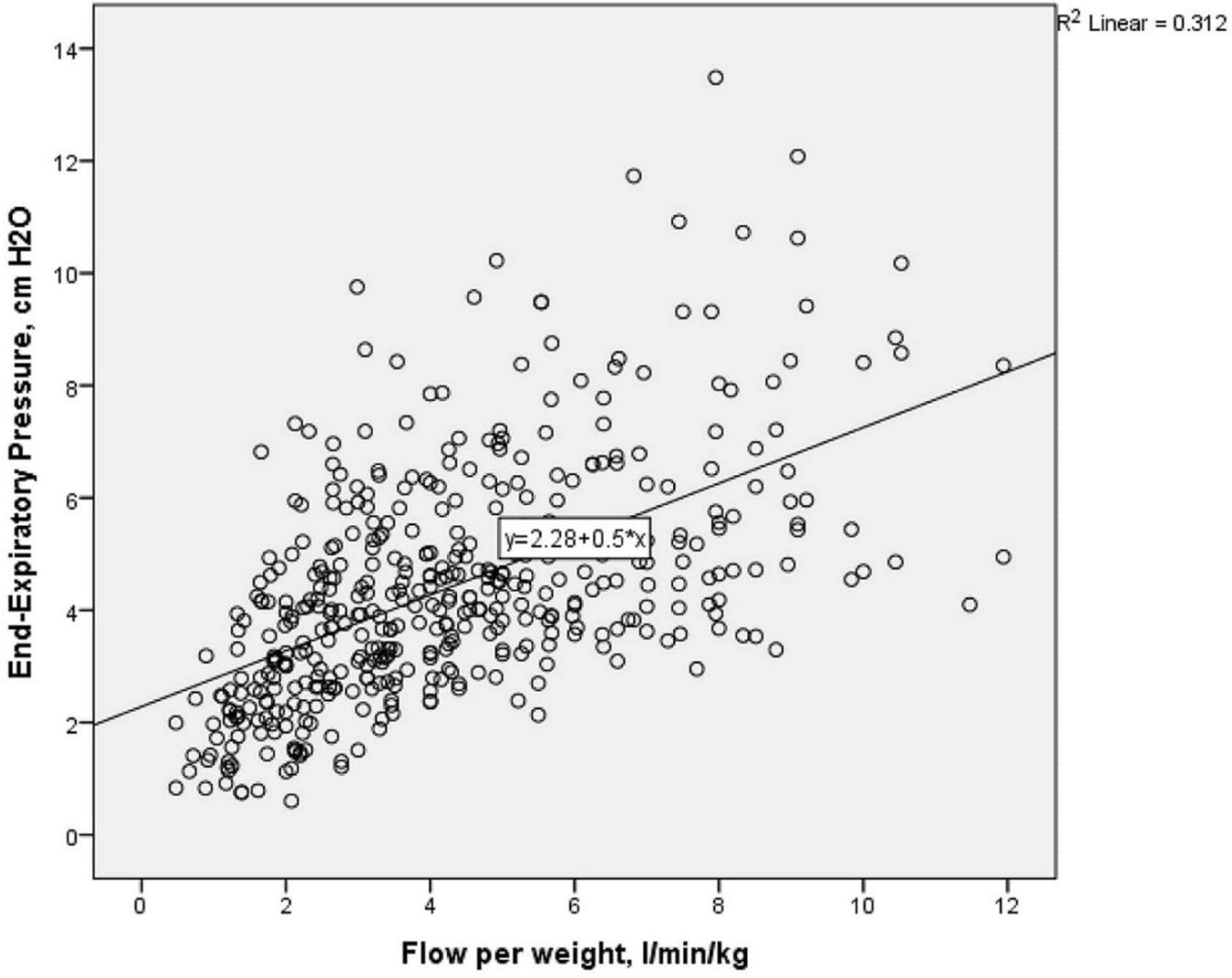
The pharyngeal pressures as shown in the table don’t clearly show the variability in the pressures obtained, which are better demonstrated in their graph:
A flow of 8 lpm/kg produced pressures between 2 and 14 cmH2O, at 2 lpm/kg pressures were between close to 0 and 7 cmH2O.
Are there any advantages to HFNC compared to CPAP? What are the disadvantages?
As mentioned above parents seem to prefer them, they also state that their child prefers them. In this randomized cross-over trial (Klingenberg C, et al. Patient comfort during treatment with heated humidified high flow nasal cannulae versus nasal continuous positive airway pressure: a randomised cross-over trial. Arch Dis Child Fetal Neonatal Ed. 2014;99(2):F134-7) parents rated their child’s “satisfaction” as an average of 8.6/10 compared to 6.9 for nasal CPAP, even though the PIPP scores were just about identical between the two groups.
That, I think, is an important difference, but must be offset by the fact that initial use of HFNC for early respiratory distress is more likely to fail than CPAP (Roberts CT, et al. Nasal High-Flow Therapy for Primary Respiratory Support in Preterm Infants. N Engl J Med. 2016;375(12):1142-51), and infants are more likely to fail extubation if they receive HFNC rather than CPAP (Uchiyama A, et al. Randomized Controlled Trial of High-Flow Nasal Cannula in Preterm Infants After Extubation. Pediatrics. 2020:e20201101. Manley BJ, et al. High-flow nasal cannulae in very preterm infants after extubation. N Engl J Med. 2013;369(15):1425-33).
This may not matter too much if you have CPAP available as a backup, but in some circumstances, failure of the HFNC might be associated with substantial pulmonary de-recruitment, and difficulty stabilising with CPAP.
In addition, we may be drowning the babies! (Reiner E, et al. Using heated humidified high-flow nasal cannulas for premature infants may result in an underestimated amount of water reaching the airways. Acta Paediatr. 2020), this was a study in an in vitro model so it is of limited applicability in terms of the actual numbers compared to the complex dynamics of a newborn’s upper airway, but a Heated humidified HFNC system deposited up to 44 mL of water over a 24 hour period in a feeding bottle being used as the model for the upper airway. A CPAP system with a heater wire in the inspiratory limb may well lead to less water deposition, but I don’t know that for sure and it would be interesting to know.
All of which leads to a few studies suggesting from several centres that when they started using more HFNC they had worsening pulmonary outcomes. Heath Jeffery RC, et al. Increased use of heated humidified high flow nasal cannula is associated with longer oxygen requirements. J Paediatr Child Health. 2017;53(12):1215-9. Hoffman SB, et al. Impact of High-Flow Nasal Cannula Use on Neonatal Respiratory Support Patterns and Length of Stay. Respir Care. 2016;61(10):1299-304. Multicentre databases have reported the same thing. Taha DK, et al. High Flow Nasal Cannula Use Is Associated with Increased Morbidity and Length of Hospitalization in Extremely Low Birth Weight Infants. J Pediatr. 2016;173:50-5 e1.
I actually wonder whether that may be because of the increased ease of use and apparent comfort of the babies on HFNC, which makes us less pressed to wean their support so we end up weaning more slowly and the babies are exposed to more positive pressure, potentially more oxygen, and maybe even more water droplets in the airway(?). The Cochrane review did not find any evidence of worse pulmonary outcomes with HFNC, but there really aren’t many trials comparing the long-term use over several weeks during the recovery phase of preterm lung disease compared to CPAP, so the Cochrane review doesn’t really cover that kind of usage. This small trial found no advantage of prolonged HFNC compared to CPAP for babies recovering from their RDS in terms of learning to feed Glackin SJ, et al. High flow nasal cannula versus NCPAP, duration to full oral feeds in preterm infants: a randomised controlled trial. Arch Dis Child Fetal Neonatal Ed. 2017;102(4):F329-F32.
So what are the indications for HFNC today?
- Initial respiratory support of the preterm infant? I think that very preterm babies (<32 weeks) are at higher risk of failing compared to CPAP and, as such babies are usually in a tertiary NICU with CPAP available, that should be their initial support. 32 to 35 week babies in a level 2 nursery could be managed initially with HFNC if CPAP is not easily available, but early contact with a referral centre should be instituted, in case of failure.
- Post-extubation support? This should be either CPAP for larger preterm babies or nIMV for smaller preterm babies. I don’t think HFNC is a good option for any baby immediately after extubation.
- Prolonged respiratory support? This is the one place where I think there may be a role for HFNC, parents prefer it and they see that their infants are more comfortable. I think that for the baby approaching 36 weeks, who is starting to be more interactive, if CPAP can’t be weaned off, then HFNC could be considered. The caveat is that we should have a protocol for weaning, with frequent evaluation of the baby and attempts at weaning, the dead space washout might reduce respiratory efforts in babies with low compliance or high resistance lungs, and they may therefore have less retractions and their nose wings might flap less (!), but beware being complacent about the baby who is stuck on HFNC, you may end up with more of them having respiratory support for longer.










Great review. This brings us to the pandoras-box that is the RAM-Canula. A CPAP “device” that is actually only approved (as far as I know) as a HFNC “device”. 🤷♂️
The resistance of the RAM cannulae is so high that they deliver almost no positive pressure to the airways when used as a CPAP interface. How that impacts on their role for HFNC I am not sure, I guess if 5 litres per minute are leaving the nasal openings of the device then they probably will generate the same dead space washout as other cannulae and the same pressures which will depend on their fit in the nostrils, mouth open or closed etc.
RAM NC does transmit pressure. But, it is lower, just like with any other nasal interface. A nice study published in JAMA answers a lot of questions. JAMA Pediatr. doi:10.1001/jamapediatrics.2020.3579
Published online November 9, 2020.
Very recently, Medtronics received FDA approval for their updated software which actually measures at the tip of the RAM NC or binasal prongs and one could characterize the nasal interface before putting on a baby and see pressures at the wye as well as at the tip of the prongs/cannula, that is, what the patient sees. One could also set ventilator settings based on PIP/PEEP measured at the wye or at the tip of the nasal interface, all of which are displayed on the ventilator. This software takes away the guess work. Be well Keith.