A couple of recent published trials have made me wonder about that question, and how to assess if an impact suggested by the results of a trial might be relevant to how I practice, and would likely be reproduced if we introduced the intervention in our NICU.
The specific question raised by these two studies is, “When the control group of a trial has an adverse outcome more frequently that I see in my practice, can I expect the same relative effect if I apply the intervention to my patients”?
For example, in this randomized controlled trial of elevated midline head positioning of extremely preterm infants, (Kochan M, et al. Elevated midline head positioning of extremely low birth weight infants: effects on cardiopulmonary function and the incidence of periventricular-intraventricular hemorrhage. J Perinatol. 2019;39(1):54-62) the authors examined the impact on IVH of keeping infants less than 1000g birth weight in a seat at 30 degress of elevation with the head maintained in a midline position during the first 4 days of life, compared to supine positioning (without elevation) accompanied by changes in head position. The primary outcome of that study was the total frequency of peri- and intra-ventricular hemorrhage, which was actually a little higher with the head elevated, 34/90, compared to 31 /90 for the FLAT group. On secondary outcome analysis the distribution of PIVH was different, with more intracerebral (grade 4) bleeds in the FLAT group than in the ELEVated group, 14 vs 6 (the incidence of grades 3 and 4 together, the more commonly used outcome, was 18 vs 11, or 20% vs 12%).
Which looks like an interesting difference if it can be confirmed. Lets assume for the moment that this is a real impact of the intervention, and not due to other effects, such as random differences (very likely in a small study), lack of blinding (impossible to do in a study like this), adverse impacts of the control intervention (possible, but a quite standard nursing approach is described), unmasked randomization (method of randomization is not adequately described apart from the “use of a randomization table”), baseline imbalance (there are some differences between groups) and other possible sources of bias. If we assume those things for the sake of this argument, we are still left in the controls with a very high incidence of intracerebral hemorrhage, 16%, among an otherwise unselected group of infants with a birth weight below 1000g, average 732 grams, average gestation 25 – 26 weeks, and an even higher incidence of grade 3 and 4 together of 20%.
In my practice, and in the CNN overall in recent years (annual reports are available on the website), the frequency of severe IVH (3+4) under 1000g is more like 11%. In the methods section of this article the authors note a recent incidence of overall IVH of 40% in their practice, and that “eighteen percent of these infants” had grades 3 and 4. It isn’t clear to me whether that means 18% of the ELBW, or 18% of the 40% who had IVH, which would be 7.2%.
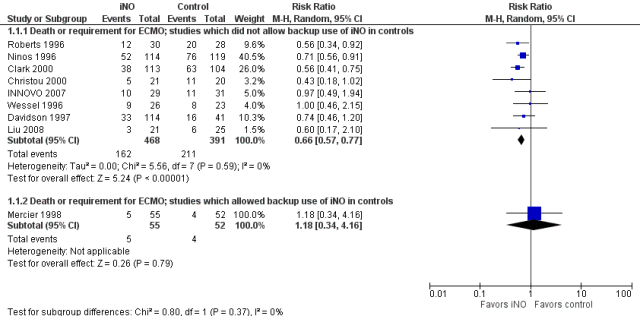
In general, to be confident about the impact of an intervention, it is preferable to see at least one very large trial with narrow confidence intervals including babies from multiple centers. The alternative being several trials with different risks in the controls and similar relative risk reductions in order to conclude that something which appears effective in a very high risk population is also effective in a lower risk population. That is the kind of information that we have for inhaled nitric oxide in term infants, for example. Here is the Forest plot for the iNO studies, as you can see, the larger studies had relative risks of “death or ECMO” between 0.56 and 0.74, and the confidence intervals all overlap substantially, despite differing control group risks.
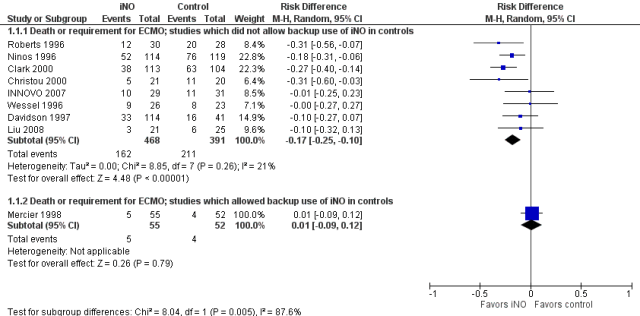
The second figure is revised from the the term iNO Cochrane review, I have ordered the studies by the control group risk of adverse outcome, and shown the risk difference. As you might expect, the higher the control group risk, the greater, in general, is the risk difference, even though the relative risk, (or risk ratio) is similar. That is why you need to be sceptical when you hear statements such as “it has only been shown to work in high-risk groups”, as we used to hear about probiotics. It is much harder to show a “statistically significant” outcome when the baseline risk is lower. But if the relative risk reductions are similar across studies with different baseline risk, that is a good reason to think that the impacts are the same.
For this positioning intervention, which has significant impacts on care of these babies, the high incidence of PIVH in the controls, reducing to an incidence with the intervention which is more similar to other recent publications, is consistent with an impact of the intervention, but is also consistent with a randomly higher incidence in the controls than usual. This is why to me the meaning of the ‘18% of these infants’ is important, if, over several years, they have had an 18% incidence of severe IVH among the ELBW, and in this study it is similar at 20%, with a substantially lower incidence in the group who had the intervention, I think that is more likely that the reduction is a real impact of the intervention, (in contrast to having a usual rate of 7.2% that increased to 20% during the period of the study).
So what do other trials say? Well there aren’t any, I think. The 2017 Cochrane review found 2 trials comparing supine positioning with the head midline, to supine with the head turned to the side (total n=110), and found no differences of note in any outcome.
A potentially important study, which certainly needs to be repeated, and it would be nice to study head elevation and midline positioning as separate interventions.
The other trial that has made me think about this issue is the PREMILOC trial. (Baud O, et al. Effect of early low-dose hydrocortisone on survival without bronchopulmonary dysplasia in extremely preterm infants (PREMILOC): a double-blind, placebo-controlled, multicentre, randomised trial. The Lancet. 2016;387(10030):1827-36). In that trial, routine administration of hydrocortisone at low doses over a total of 10 days led to a reduction in mortality before discharge, and a reduction in oxygen use at 36 weeks compared to control in infants of 24 to under 28 weeks.
When trying to assess the relevance for my practice, I note that the study did not include any infants under 24 weeks, or with a birth weight below the 3rd percentile, or with ruptured membranes before 22 weeks, or with 5 minute Apgar under 4, or with congential anomalies detected prenatally. It was overall a very high quality multicenter trial including a little over 1000 babies.
One of the striking things in the results of that trial is the high mortality before discharge in the 24 and 25 week infants, which is 44% with placebo and 42% with hydrocortisone. In my practice and in the 2017 CNN report, mortality among infants at 24 and 25 weeks admitted to the NICU (data which include those other very high risk groups) is 20%. The incidence of severe IVH is also very high in PREMILOC at about 25%. Even at 26 and 27 weeks mortality in both groups is higher than the CNN mortality, 7.6% HC and 15.3% placebo in the PREMILOC trial, compared to 6% in the CNN, again including growth restricted babies.
There are also other signs in the publication that the treatment approaches were substantially different to what we do in our practice, 35% of placebo and 30% of hydrocortisone babies were on inotropes at study entry, 43% in each group received insulin during the trial.
If I already have a mortality which is substantially lower than the intervention group in a trial such as this, am I likely to see an impact of the intervention in my practice?
The answer, I would say, is definitely a ‘maybe’. If the same pathophysiology for the adverse outcome exists, and if the underlying risk factors are similar (including aspects of obstetric management), then I might expect a similar relative reduction in mortality, and a smaller absolute reduction in mortality. Of course as the absolute benefit become smaller, the potential secondary adverse effects of the intervention become potentially more important, in this specific case the increase in late-onset sepsis may be more important in the calculation of the risk-benefit balance.
I am not saying that we should ignore the results of trials if there are details of management or outcomes that are different to local data, that would just lead to chaos and a complete rejection of evidence-based practice, I am saying that we need to be thoughtful about interpretation and application of trial data. If local approaches and outcomes are dramatically different from a published trial, the we should take into account the toxicity/adverse impacts of the intervention, and whether there are other data from a variety of populations that are consistent. To go back to the probiotics example, the relative reduction in the risk of NEC is similar across studies, suggesting a real impact overall, although there are differences between the studies they consistently found no adverse effects of priobiotic use. Even if you have locally a low incidence of NEC, it is likely that it will be further reduced by probiotics, without any increased risk, and without much cost.
For routine midline head-elevated positioning, or routine use of hydrocortisone in the 1st day of life, I will wait for more data from different groups with differing baseline risks before changing my practice.









Or as some have commented, it is hard to improve upon zero.