Ybarra M, et al. Low-Grade Germinal Matrix Hemorrhage-Intraventricular Hemorrhage and Concomitant Preterm Brain Injuries: Neurodevelopmental Outcomes at 3 Years of Age. J Pediatr. 2025:114713. Previous studies of the long term outcome of infants with germinal matrix or low grade IVH have been inconsistent. Some have shown an association with poorer developmental progress, and others have shown no impact. Some of this variability may be due to uncertainty about diagnostic criteria, with slightly larger amounts of intraventricular blood being classified differently. Some is probably due to the variable association with other brain injury, not readily seen on ultrasound, such as white matter injury, or cerebellar haemorrhages. We now routinely perform imaging of the posterior fossa, which was not easy with older ultrasound machines, but small cerebellar haemorrhages are still hard to see, without MRI.
In this cohort from Toronto, 175 infants <32 weeks GA had ultrasounds, they also had early cerebral MRI at 32 to 34 weeks, if they were stable, and then again at term equivalent age. Neurologic and developmental assessments were performed at 3 years (Bayley version III). As for the results, low grade haemorrhages had no correlation with outcomes, unless associated with either large cerebellar haemorrhages or more extensive white matter injury. It has always been fascinating to me that germinal matrix haemorrhage, which destroys the primary source of cortical neurones, has so little impact on long term outcomes. It speaks to the plasticity of the newborn brain, if the Germinal Matrix is injured, other parts of the brain take over neurone production.
Take home message : there was no apparent impact of GMH or small intraventricular haemorrhages without dilatation on long term development. Cerebellar haemorrhages, if large, are associated with delayed language development at 3 years, and white matter abnormalities, if extensive, are associated with motor delay, and cerebral palsy.

Interesting review article on the impact of pasteurization, using the standard (Holder) pasteurization method, as well as some information about alternatives. The dash (-) in the figure above means no effect, rather than deletion. As you can see there are multiple impacts of pasteurization, as well as the expected impact on bacteria, some bacteria are resistant to Holder pasteurization, so donor breast milk still has an impact on the preterm intestinal microbiome. Both by direct colonization with the surviving organisms, but also because of the impact of HMOs and other components of human milk which remain despite pasteurization.
The figure also shows, in the upper right third, some alternative pasteurization methods which have been investigated, and which all show lesser impacts on breast milk components, HTST (high temperature short treatment) HPP (high pressure pasteurization) and UV-C (UV-C!). These alternative methods are equally effective at reducing bacterial load in the donor milk, and hopefully can be used in the future to give donor milk which is closer to Mothers Own Milk.
Take home message : Holder Pasteurization has major impacts on the composition of human milk. Alternative methods should be investigated, and approved.
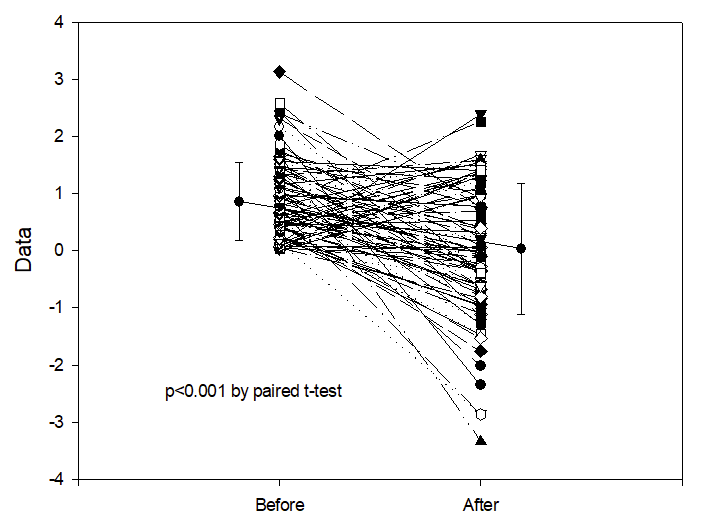
In this observational study, the authors correlated the diet of a cohort of preterm infants <32 weeks GA with the findings on MRI at term. The cohort was enrolled over a long period, including a couple of years prior to the availability in their centre of donor milk (DHM) 2012-2014, and several years afterward 2014-2022. They include babies who almost exclusively received Mother’s own milk (MoM) and those receiving mostly formula, as well as the group with DHM. Brain volumes were greater in the human milk groups compared to formula, and diffusion tensor imaging showed diffusivity differences also, in the Corpus Callosum and the PLIC (posterior limb of the internal capsule). As the authors note, there is no good evidence from RCTs that DHM leads to better clinical neurological or developmental outcomes than formula. Nevertheless, these data are consistent with a beneficial effect of human milk on brain development, shared by DHM, and MoM.
Take home message : human milk seems to promote larger brains.
One of the benefits of MoM is that it routinely contains probiotic organisms, usually including Bifidobacteria. In this trial, 70 preterm infants <32 weeks were randomized to control or to a supplement of Bifidobacterium animalis susp lactis. As often happens in some journals the article is written in somewhat strange English; one example : “Quality control and data analysis were conducted after instrument analysis, using assessment of the peak significante equation of standard curves”. They ran a statistical comparison of the baseline characteristics of the randomized groups. This is a practice that Pediatric Research should know is ridiculous. If the groups were randomized, why run such a statistical test? It is superfluous, potentially misleading, and the CONSORT statement specifically states that it should not be done, Pediatric Research is supposed to follow CONSORT guidelines.
I started to include this article in the post as I thought it was a demonstration of the possible anti-inflammatory impact of this Bifidobacterium on the preterm intestine. But I now realize that I haven’t got a clue what most of it means. This following figure for example, is supposed to show correlations between a large number of “metabolites”; about 30 were selected from over 250 that were found in the stools, including, for example, 34 different bile acids. These figures are supposed to show correlations, negative and positive, between “metabolites”.

The legend to the figure states “Red indicates positive correlation, blue indicates negative correlation, and the darker the colour, the stronger the correlation. a Probiotic group week 2 VS Control group week 2. b Probiotic group week 2 VS Control group week 2″…. What on earth is this supposed to mean? Aside from the fact that the potential of at least 62,500 comparisons were possible, is this comparison within the 2 groups, or between the 2 groups?
What it seems to show is that they measured a huge number of molecules, the concentrations of some of them were correlated with the concentrations of others. But so what?
They also show dramatic differences in serum TLR4 concentrations between groups. I am unsure if circulating concentrations of TLR4 are of any interest; TLR4 is normally attached to granulocytes, as part of the receptor complex which recognizes lipopolysaccharide. Nevertheless, published serum concentrations range between the pg/mL range to the mg/mL range, with these new results being intermediate in the ng/mL range. Such enormously variant normal ranges (over 1 million fold differences) make me very sceptical about any results. Serum TNF-α and IL-Iß were also dramatically lower in the Bifido group. They also give exactly the same data, regarding clinical complications, in table 3 and a completely superfluous figure 10.
Pediatric Research used to be a journal that only published high-quality research, although they were rarely clinical studies, which was previously one of my criticisms of the journal. If this is typical of the quality of what is currently getting through peer review and editorial control, then the journal has fallen far indeed.
Take home message : Pediatric Research is no longer the high-quality source it once was.
This is a report of the 3 year outcomes of babies from a cluster RCT of a quality control initiative in Japan. The original publication showed no impact of the QI program (INTACT), so the authors combined the groups for this publication describing their neurological and developmental outcomes. Babies were VLBW and ranged from 22 to 31 weeks GA. Below is a selection from the extensive results, “severely delayed” refers to being <70 on the cognitive subscale of the Kyoto Scales of Psychological Development. The KSPD seems to have a similar mean to the BSID ver3, when tested on the same infants, but have a wider distribution, so a score <70 was considered severely delayed.



There was very little blindness or deafness, so, as usual, it was cognitive delay which was responsible for most of the infants who were classified as “NDI”. Unfortunately, the authors don’t report many things which matter to parents, in particular there is no mention of behavioural problems. They do have a table that they call “functional outcomes” but that is actually a report of the medical interventions being received by the infants, at 3 years of age, including home oxygen, NG tube feeding, anticonvulsant medications, etc. All of which were rare.
Take home message : The majority of survivors at every gestational age, even the most immature, do not have “moderate or severe NDI”. There is a progressive increase in “moderate or severe NDI” as GA decreases.